Advertisements
Advertisements
Questions
How do you convert the following:
Ethanol to propanenitrile
How the following conversion can be carried out?
Ethanol to propanenitrile
Advertisements
Solution 1
\[\ce{\underset{Ethanol}{CH3 - CH2 - OH} ->[red P/Br2] \underset{Bromoethane}{CH3 - CH2 - Br} ->[KCN, aq.ethanol]\underset{Propanenitrile}{CH3 - CH2 - CN}}\]
Solution 2
\[\ce{\underset{Ethanol}{CH3CH2OH} ->[P/I2, \Delta] \underset{Iodoethane}{CH3CH2I} ->[KCN/EtOH-H2O][nucleophilic substitution] \underset{Propanenitrile}{CH3CH2CN}}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
RELATED QUESTIONS
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Identify 'A' in the following reaction -
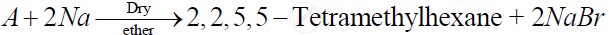
(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Optically active isomers but not mirror images are called ____________.
Which of the following compounds is optically active?
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
The increasing order of nucleophilicity would be:
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.
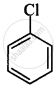
(I)
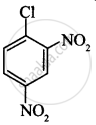
(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 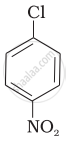 |
| (II) | 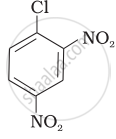 |
| (III) | 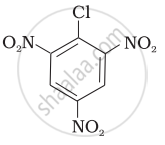 |
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
CCl4 is insoluble in water because:-
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Acetic anhydride from acetic acid
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
