Advertisements
Advertisements
Question
State and explain Markownikoff's rule with suitable example
Solution
The Markovnikov's rule states that when to an unsymmetrical alkene a reagent is added then the negative part of the addendum (adding molecule) gets attached to that carbon atom which possesses lesser number of hydrogen atoms. For example
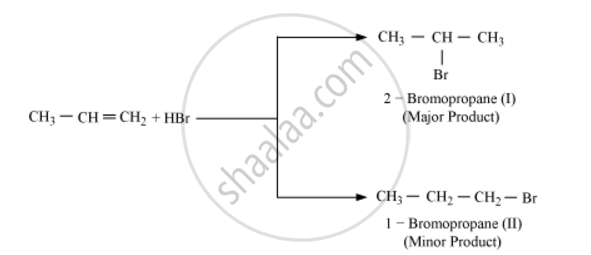
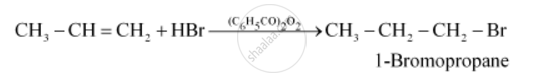
The anti- Markovnikov's rule is the reverse of the above and states that when to an unsymmetrical alkene a reagent is added then the negative part of the addendum (adding molecule) gets attached to that carbon atom which possesses more number of hydrogen atoms. It is normally followed when hydrogen and many other peroxide is added to a reaction system. Thus when the reaction system contains peroxides, anti- Markovnikov's rule will be followed. If any particular reagent has not been mentioned, then Markovnikov's rule will be followed. Example, in the following reaction anti-Markovnikov's rule is followed due to the presence of peroxide
APPEARS IN
RELATED QUESTIONS
Identify the product ‘D’ in the following sequence of reactions:
\[\ce{H3C - CH2 - CH2 - Cl \underset{KOH}{\overset{Alc}{->}} 'B' \overset{HBr}{->} 'C' \underset{Elther}{\overset{Na}{->}}'D'}\]
Identify ‘A’ and ‘B’ in the following reaction :
\[\ce{CH3 - CH = CH2 ->[HBr]'A' ->[alc.KOH]'B'}\]
How do you convert: 2-bromobutane to but-2-ene
Write the main products when n-butyl chloride is treated with alcoholic KOH.
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
1-Bromo-1-methylcyclohexane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2-Chloro-2-methylbutane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2, 2, 3-Trimethyl-3-bromopentane
How will you bring about the following conversion?
But-1-ene to but-2-ene
Write the structure of the major organic product in the following reaction:
\[\ce{(CH3)3CBr + KOH ->[ethanol][heat]}\]
How the following conversion can be carried out?
1-Bromopropane to 2-bromopropane
How the following conversion can be carried out?
2-Chloropropane to 1-propanol
How the following conversion can be carried out?
2-Bromopropane to 1-bromopropane
Draw a neat, labelled energy profile diagram for SN1 reaction mechanism.
What are racemates?
Observe the following compounds and answer the questions given below.
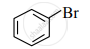
(I)
\[\ce{\underset{\text{(II)}}{CH3 - CH2 - Br}}\]
- Identify the type of halides.
- Explain the nature of the C – Br bond in both of these halides.
- Which of these compounds will undergo aqueous alkaline hydrolysis readily? Write the reaction in support of your answer.

'A' is:
Deamination of meso- di bromobutane gives mainly:-
Identify the major product formed when 2-cyclohexylchloroethane undergoes a dehydrohalogenation reaction. Name the reagent which is used to carry out the reaction.
Elimination of bromine from 2-bromobutane results in the formation of ______.
The conversion of an alkyl halide into an alkene by alcoholic KOH is classified as ______.
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3 C(CI) (C2H5)CH2CH3 }\]
