Advertisements
Advertisements
प्रश्न
Comment on the following statement: the temperature of all the molecules in a sample of a gas is the same.
उत्तर
Yes, at equilibrium all the molecules in a sample of gas have the same temperature. This is because temperature is defined as the average kinetic energy for all the molecules in a system. Since all the molecules have the same average, temperature will be the same for all the molecules.
APPEARS IN
संबंधित प्रश्न
When we place a gas cylinder on a van and the van moves, does the kinetic energy of the molecules increase? Does the temperature increase?
Do you expect the gas in a cooking gas cylinder to obey the ideal gas equation?
A gas is kept in an enclosure. The pressure of the gas is reduced by pumping out some gas. Will the temperature of the gas decrease by Charles's low?
Find the number of molecules of an ideal gas in a volume of 1.000 cm3 at STP.
An ideal gas is kept in a long cylindrical vessel fitted with a frictionless piston of cross-sectional area 10 cm2 and weight 1 kg in figure. The vessel itself is kept in a big chamber containing air at atmospheric pressure 100 kPa. The length of the gas column is 20 cm. If the chamber is now completely evacuated by an exhaust pump, what will be the length of the gas column? Assume the temperature to remain constant throughout the process.

When a gas is heated, its temperature increases. Explain this phenomenon on the basis of the kinetic theory of gases.
Calculate the ratio of the mean square speeds of molecules of a gas at 30 K and 120 K.
The emissive power of a sphere of area 0.02 m2 is 0.5 kcal s-1m-2. What is the amount of heat radiated by the spherical surface in 20 seconds?
Compare the rates of emission of heat by a blackbody maintained at 727°C and at 227°C, if the black bodies are surrounded by an enclosure (black) at 27°C. What would be the ratio of their rates of loss of heat?
The number of degrees of freedom, for the vibrational motion of a polyatomic molecule, depends on the ______
Why the temperature of all bodies remains constant at room temperature?
If the density of nitrogen is 1.25 kg/m3 at a pressure of 105 Pa, find the root mean square velocity of nitrogen molecules.
Compare the rate of radiation of metal bodies at 727 °C and 227 °C.
What is the microscopic origin of temperature?
Explain in detail the kinetic interpretation of temperature.
The graph of kinetic energy against the frequency v of incident light is as shown in the figure. The slope of the graph and intercept on X-axis respectively are ______.

Assuming the expression for the pressure exerted by the gas on the wall of the container, it can be shown that pressure is ______.
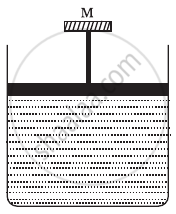
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased ______.
A gas mixture consists of molecules of types A, B and C with masses mA > mB > mC. Rank the three types of molecules in decreasing order of average K.E.
If a = 0. 72 and t = 0.04, then the value of r is ______.
