Advertisements
Advertisements
प्रश्न
The decay constant of 238U is 4.9 × 10−18 S−1. (a) What is the average-life of 238U? (b) What is the half-life of 238U? (c) By what factor does the activity of a 238U sample decrease in 9 × 109 years?
उत्तर
Given:
Decay constant, `lambda = 4.9 xx 10^-18 "s"^-1`
(a) Average life of uranium (`tau`) is given by
`tau = 1/lambda`
= `1/(4.9 xx 10^-18)`
= `1/4.9 xx 10^18 "s"`
= `10^16/(4.9 xx 365 xx 24 xx 36) "years"`
= `10^16/(4.9 xx 365 xx 24 xx 36) "years"`
= `6.47 xx 10^-7 xx 10^16 "years"`
= `6.47 xx 10^9 "years"`
(b) Half-life of uranium (`T_"1/2"`) is given by
`T_"1/2" = 0.693/lambda = 0.693/(4.9 xx 10^-18)`
= `0.693/4.9 xx 10^18 "s"`
= `0.1414 xx 10^18 "s"`
= `(0.1414 xx 10^18)/(365 xx 24 xx 3600)`
= `(1414 xx 10^12)/(365 xx 24 xx 36)`
= `4.48 xx 10^-3 xx 10^12`
= `4.5 xx 10^9` years
(c) Time, t = 9 × 109 years
Activity (A) of the sample, at any time t, is given by
`A = A_0/2^(t/T_"1/2")`
Here , `A_0` = Activity of the sample at t = 0
`therefore A_0/A = 2^((9 xx 10^9)/(4.5 xx 10^9)) = 2^2 = 4`
APPEARS IN
संबंधित प्रश्न
Derive the mathematical expression for law of radioactive decay for a sample of a radioactive nucleus
Write symbolically the process expressing the β+ decay of `""_11^22Na`. Also write the basic nuclear process underlying this decay.
The half-life of `""_38^90 "Sr"` is 28 years. What is the disintegration rate of 15 mg of this isotope?
Using the equation `N = N_0e^(-lambdat)` obtain the relation between half-life (T) and decay constant (`lambda`) of a radioactive substance.
(a) Derive the relation between the decay constant and half life of a radioactive substance.
(b) A radioactive element reduces to 25% of its initial mass in 1000 years. Find its half life.
Define 'activity' of a radioactive substance ?
Define the activity of a given radioactive substance. Write its S.I. unit.
A radioactive nucleus ‘A’ undergoes a series of decays according to the following scheme:
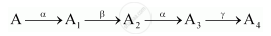
The mass number and atomic number of A are 180 and 72 respectively. What are these numbers for A4?
In a radioactive decay, neither the atomic number nor the mass number changes. Which of the following particles is emitted in the decay?
Lithium (Z = 3) has two stable isotopes 6Li and 7Li. When neutrons are bombarded on lithium sample, electrons and α-particles are ejected. Write down the nuclear process taking place.
The half-life of 40K is 1.30 × 109 y. A sample of 1.00 g of pure KCI gives 160 counts s−1. Calculate the relative abundance of 40K (fraction of 40K present) in natural potassium.
A radioactive substance disintegrates into two types of daughter nuclei, one type with disintegration constant λ1 and the other type with disintegration constant λ2 . Determine the half-life of the radioactive substance.
What is the amount of \[\ce{_27^60Co}\] necessary to provide a radioactive source of strength 10.0 mCi, its half-life being 5.3 years?
Before the year 1900 the activity per unit mass of atmospheric carbon due to the presence of 14C averaged about 0.255 Bq per gram of carbon.
(a) What fraction of carbon atoms were 14C?
(b) An archaeological specimen containing 500 mg of carbon, shows 174 decays in one hour. What is the age of the specimen, assuming that its activity per unit mass of carbon when the specimen died was equal to the average value of the air? The half-life of 14C is 5730 years.
What percentage of radioactive substance is left after five half-lives?
The half-life of a radioactive nuclide is 20 hrs. The fraction of the original activity that will remain after 40 hrs is ______.
If 10% of a radioactive material decay in 5 days, then the amount of original material left after 20 days is approximately :
Suppose we consider a large number of containers each containing initially 10000 atoms of a radioactive material with a half life of 1 year. After 1 year ______.
Samples of two radioactive nuclides A and B are taken. λA and λB are the disintegration constants of A and B respectively. In which of the following cases, the two samples can simultaneously have the same decay rate at any time?
- Initial rate of decay of A is twice the initial rate of decay of B and λA = λB.
- Initial rate of decay of A is twice the initial rate of decay of B and λA > λB.
- Initial rate of decay of B is twice the initial rate of decay of A and λA > λB.
- Initial rate of decay of B is the same as the rate of decay of A at t = 2h and λB < λA.
