Advertisements
Advertisements
Question
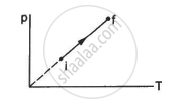
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
Options
is positive
is negative
is zero
cannot be obtained from this information
Solution
is zero
Work done by the gas during the process,
ΔW = P Δ V
Here,
P = Pressure
ΔV = change in volume
Since the process described in the figure is isochoric, P = kT. As volume remains constant (ΔV = 0), ΔW = 0.
APPEARS IN
RELATED QUESTIONS
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if heat is added to it?
Should the internal energy of a system necessarily increase if its temperature is increased?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
When a tyre bursts, the air coming out is cooler than the surrounding air. Explain.
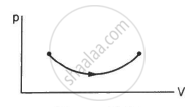
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
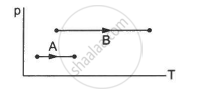
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder ______________ .
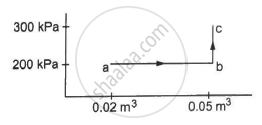
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.
A gas is initially at a pressure of 100 kPa and its volume is 2.0 m3. Its pressure is kept constant and the volume is changed from 2.0 m3 to 2.5 m3. Its Volume is now kept constant and the pressure is increased from 100 kPa to 200 kPa. The gas is brought back to its initial state, the pressure varying linearly with its volume. (a) Whether the heat is supplied to or extracted from the gas in the complete cycle? (b) How much heat was supplied or extracted?
What is the energy associated with the random, disordered motion of the molecules of a system called as?
Define heat.
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
When does a system lose energy to its surroundings and its internal energy decreases?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
In thermodynamics, heat and work are ______.
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
