Advertisements
Advertisements
Question
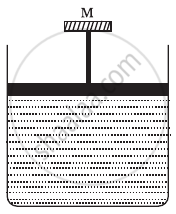
An ideal gas is kept in a long cylindrical vessel fitted with a frictionless piston of cross-sectional area 10 cm2 and weight 1 kg in figure. The vessel itself is kept in a big chamber containing air at atmospheric pressure 100 kPa. The length of the gas column is 20 cm. If the chamber is now completely evacuated by an exhaust pump, what will be the length of the gas column? Assume the temperature to remain constant throughout the process.

Solution
Atmospheric pressure inside the cylinderical vessel, `P_0 = 10^5 "Pa"`
A = 10 cm2 = 10 × 10-4 m2
Pressure due to the weight of the piston `= (mg)/A =( 1 × 9.8 ) / (10×10^-4)`
`P_1 = 10^5+9.8×10^3`
`V_1 = 0.2×10×10^-4 = 2×10^-4`
After evacution , external pressure above the piston = 0
`P_2 = 0+9.8×10^3`
Now,
`P_1V_1 = P_2V_2`
Let L be the final length of the gas column . Then,
`V_2 = 10×10^-4L`
`rArr(10^5+9.8×10^3) × 0.2×10×10^-4 = 9.8 × 10^3 × 10 × 10^-4"L"`
L = 2.2 m
APPEARS IN
RELATED QUESTIONS
The pressure of an ideal gas is written as \[P = \frac{2E}{3V}\] . Here E refers to
Which of the following quantities is the same for all ideal gases at the same temperature?
(a) The kinetic energy of 1 mole
(b) The kinetic energy of 1 g
(c) The number of molecules in 1 mole
(d) The number of molecules in 1 g
The temperature and pressure at Simla are 15.0°C and 72.0 cm of mercury and at Kalka these are 35.0°C and 76.0 cm of mercury. Find the ratio of air density at Kalka to the air density at Simla.
Use R=8.314J K-1 mol-1
At what temperature the mean speed of the molecules of hydrogen gas equals the escape speed from the earth?
Use R = 8.314 JK-1 mol-1
Figure shows a cylindrical tube of cross-sectional area A fitted with two frictionless pistons. The pistons are connected to each other by a metallic wire. Initially, the temperature of the gas is T0 and its pressure is p0 which equals the atmospheric pressure. (a) What is the tension in the wire? (b) What will be the tension if the temperature is increased to 2T0 ?

The weather report reads, "Temperature 20°C : Relative humidity 100%". What is the dew point?
The temperature and the dew point in an open room are 20°C and 10°C. If the room temperature drops to 15°C, what will be the new dew point?
Answer in brief:
A gas in a cylinder is at pressure P. If the masses of all the molecules are made one-third of their original value and their speeds are doubled, then find the resultant pressure.
Answer in brief:
Show that rms velocity of an oxygen molecule is `sqrt2` times that of a sulfur dioxide molecule at S.T.P.
Explain, on the basis of the kinetic theory of gases, how the pressure of a gas changes if its volume is reduced at a constant temperature.
Find the kinetic energy of 5 litres of a gas at STP, given the standard pressure is 1.013 × 105 N/m2.
Why the temperature of all bodies remains constant at room temperature?
Explain in detail the kinetic interpretation of temperature.
The average K.E. of hydrogen molecules at 27° C is E. The average K.E. at 627° C is ____________.
A ring of mass m and radius r rotates about an axis passing through its centre and perpendicular to its plane with angular velocity `omega`. Its kinetic energy is ______.
Assuming the expression for the pressure exerted by the gas on the wall of the container, it can be shown that pressure is ______.
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased ______.
The molecules of a given mass of a gas have root mean square speeds of 100 ms−1 at 27°C and 1.00 atmospheric pressure. What will be the root mean square speeds of the molecules of the gas at 127°C and 2.0 atmospheric pressure?
Assuming the expression for the pressure P exerted by an ideal gas, prove that the kinetic energy per unit volume of the gas is `3/2` P.
