Topics
Force, Work, Power and Energy
Force
Light
Work, Energy and Power
- Concept of Work
- Measurement of Work
- Expression for Work (W = F S cosθ)
- Work Done by the Force of Gravity (W = mgh)
- Concept of Power
- Work vs Power
- Concept of Energy
- Energy vs Power
- Mechanical Energy > Potential Energy (U)
- Mechanical Energy > Kinetic Energy (K)
- Potential vs Kinetic Energy
- Conversion of Potential Energy into Kinetic Energy
- Forms of Energy > Solar Energy
- Forms of Energy > Heat Energy
- Forms of Energy > Light Energy
- Forms of Energy > Chemical Energy
- Forms of Energy > Hydro Energy
- Forms of Energy > Electrical Energy
- Forms of Energy > Nuclear Energy
- Forms of Energy > Geo Thermal Energy
- Forms of Energy > Wind Energy
- Forms of Energy > Sound Energy
- Forms of Energy > Magnetic Energy
- Forms of Energy > Mechanical Energy
- Conversion of Energies
- Principle of Conservation of Energy
- Proof: Kinetic + Potential Energy = Constant for Free Fall
- Application of the Principle of Conservation of Energy
Sound
Machines
- Concept of Machines
- Technical Terms Related to a Machine
- Principle of a Machine
- Efficiency, Mechanical Advantage, and Velocity Ratio
- Levers
- Types of Levers
- Pulley
- Single Fixed Pulley
- A Single Movable Pulley
- Single Pulley vs Single Movable Pulley
- Combination of Pulleys
- Using one fixed pulley and other movable pulleys
- Using several pulleys in two blocks (block and tackle system)
Electricity and Magnetism
Refraction of Light at Plane Surfaces
- Light: Reflection and Refraction
- Refraction of Light
- Laws of Refraction
- Speed of Light in Different Media
- Principle of Reversibility of the Path of Light
- Refraction Laws & Glass Index
- Refraction of Light Through a Rectangular Glass Block
- Multiple Images in a Thick Mirror
- Prism
- Refraction of Light Through a Prism
- Real and Apparent Depth
- Apparent Bending of a Stick Under Water
- Consequences of Refraction of Light
- Transmission of Light From a Denser Medium to a Rarer Medium
- Critical Angle
- Relationship between Critical Angle and Refractive Index
- Total Internal Reflection
- Total Internal Reflection in a Prism
- Total Internal Reflection Through a Right-Angled Isosceles Prism
- Total Internal Reflection Through an Equilateral Prism
- Total Internal Reflection Through Right-angled prism
- Use of a Total Internal Reflecting Prism in Place of a Plane Mirror
- Total Internal Reflection vs Reflecting from a Plane Mirror
- Consequences of Total Internal Refraction
Heat
Refraction Through a Lens
- Concept of Lenses
- Action of a Lens as a Set of Prisms
- Technical Terms Related to a Lens
- Convex Lens vs Concave Lens
- Refraction of Light Through an Equi-Convex Lens and an Equi-Concave Lens
- Principal Rays for Ray Diagrams
- Real Image vs Virtual Image
- Construction of a Ray Diagram for a Lens
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Linear Magnification
- Power of a Lens
- Magnifying Glass Or Simple Microscope
- Application of Lenses
- Experimental Determination of Focal Length of Convex Lens
- Convex Lens vs Concave Lens
Modern Physics
Spectrum
- Deviation Produced by a Triangular Prism
- Colour in White Light with Their Wavelength and Frequency Range
- Dispersion of Light
- Electromagnetic Spectrum
- Properties and Uses of Different Radiations of the Electromagnetic Spectrum
- Distinction between Ultraviolet, Visible, and Infrared Radiations
- Scattering of Light
- Applications of Scattering of Light
Sound
- Sound Waves
- Light Waves vs Sound Waves
- Reflection of Sound Waves
- Echo
- Determination of Speed of Sound by the Method of Echo
- Use of Echoes
- Natural Vibrations
- Damped Vibrations
- Natural Vibrations vs Damped Vibrations
- Forced Vibrations
- Natural Vibrations vs Forced Vibrations
- Resonance (a special case of forced vibrations)
- Demonstration of Resonance
- Forced Vibrations vs Resonant Vibrations
- Examples of Resonance
- Characteristics of Sound
- Loudness and Intensity
- Pitch and frequency
- Quality and Wave Form
- Music and Noise
Current Electricity
- Electric Charge
- Electric Current
- Potential and Potential Difference
- Electric Resistance
- Ohm's Law
- Experimental Verification of Ohm’s Law and Ohmic Resistors
- Ohmic and Non-ohmic Resistors
- Specific Resistance
- Choice of Material of a Wire
- Superconductors
- Electro-Motive Force of a Cell
- Terminal Voltage of a Cell
- Internal Resistance of a Cell
- Resistance of a System of Resistors
- Resistors in Series
- Resistors in Parallel
- A combination of resistors in both series and parallel
- Forms of Energy > Electrical Energy
- Measurement of Electrical Energy
- Electrical Power
- Commercial Unit of Electrical Energy
- Power Rating of Common Electrical Appliances
- Household Consumption of Electric Energy
- Heating Effect of Electric Current
Household Circuits
- Transmission of Power from the Power Generating Station to the Consumer
- Power Distribution to a House
- House Wiring (Ring System)
- Fuse
- Reason for connecting the fuse in the live wire
- Current Rating of a Fuse
- Switches
- Circuits with Dual Control Switches (Staircase Wire)
- Earthing
- Three-pin Plug and Socket
- Colour Coding of Wires in a Cable
- High Tension Wires
- Precautions to Be Taken While Using Electricity
Electro-Magnetism
- Oersted's Experiment
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Electromagnet
- Permanent Magnet
- Comparison of an Electro Magnet with a Permanent Magnet
- Advantages of an Electromagnet over a Permanent Magnet
- Uses of Electromagnet
- Force on a Current Carrying Conductor in a Magnetic Field
- Simple D.C. Motor
- Electromagnetic Induction
- Demonstration of the Phenomenon of Electromagnetic Induction
- Faraday's Explanation
- Faraday's Laws of Electromagnetic Induction
- A.C. Generator
- Frequency of an a.c. in Household Supplies
- Comparison Between A.C. Generator and D.C. Motor
- Transformers
Calorimetry
- Heat
- The Temperature and a Thermometer
- Factors Affecting the Quantity of Heat Absorbed to Increase the Temperature of a Body
- Heat vs Temperature
- Thermal or Heat Capacity
- Specific Heat Capacity
- Heat Capacity vs Specific Heat Capacity
- Specific Heat Capacity of Some Common Substances
- Calorimetry
- Principle of Method of Mixtures or Principle of Calorimetry
- Natural Phenomena and Consequences of High Specific Heat Capacity of Water
- Examples of High and Low Heat Capacity
- Change of State
- Melting and Freezing
- Melting Point and Its Effects
- Vaporisation or Boiling
- Boiling Point and Its Effects
- Latent Heat
- Specific Latent Heat of Fusion of Ice
- Explanation of Latent Heat of Melting based on Kinetic Model
- Natural Consequences of High Specific Latent Heat of Fusion of Ice
Radioactivity
- Structure of the Atom and Nucleus
- Atomic Models
- Isotopes
- Isobars
- Isotones
- Radioactivity
- Radioactivity as emission of Alpha, Beta, and Gamma Radiations
- Properties of Alpha Particles
- Properties of Beta Particles
- Properties of Gamma Radiations
- Distinction between the Properties of α, β, and γ Radiations
- Changes Within the Nucleus in Alpha, Beta and Gamma Emission
- Uses of Radioactive Isotopes
- Sources of Harmful Radiations
- Harmful Effects of Radiation
- Safety Precautions While Using Nuclear Energy
- Background Radiations
- Forms of Energy > Nuclear Energy
- Nuclear Fission
- Radioactive Decay Vs Nuclear Fission
- Nuclear Fusion
- Nuclear Fission Vs Nuclear Fusion
- Introduction
- Definition: Calorimetry
- Definition: Calorimeter
- Calorimeter
- The Principle of Calorimetry
- Experiment: Method of Mixtures
- Derivation of the Calorimetry Equation
- Example
- Key Points: Calorimetry
Introduction
A thermometer tells you how hot something is, but it doesn't tell you how much heat energy an object contains or exchanges. To answer that question, scientists developed a technique called calorimetry and a device called a calorimeter.
Definition: Calorimetry
Calorimetry is the science of measuring heat exchange during physical or chemical processes. The word comes from the Latin calor (heat) + Greek metron (measure).
Definition: Calorimeter
A calorimeter is a cylindrical vessel which is used to measure the amount of heat gained (or lost) by a body when it is mixed with another body or substance.
Calorimeter
A calorimeter is an insulated device used to measure the heat gained or lost by a substance during a thermal process. It works on a simple idea: trap all the heat inside the device so nothing escapes, and then measure the temperature change.
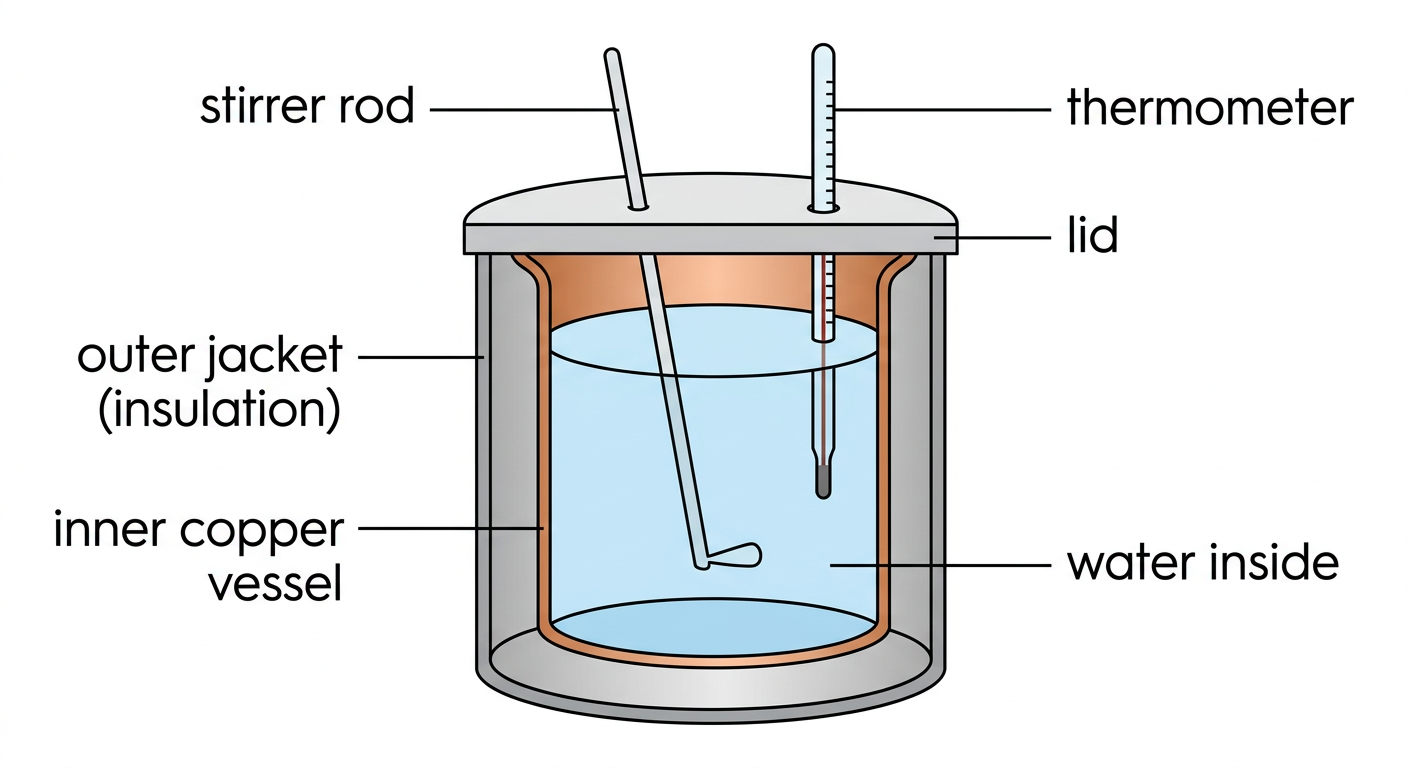
Fig.: Cross-section of a simple water calorimeter showing all key components
| Part | Material | Function |
|---|---|---|
| Inner Vessel | Copper or Aluminium | Holds water; copper is an excellent thermal conductor for quick heat exchange |
| Outer Jacket | Insulated material | Prevents heat loss to the surroundings |
| Insulating Air Gap | Air / Padding | Creates thermal isolation between inner and outer vessels |
| Stirrer | Metal (copper) | Ensures uniform temperature distribution throughout the water |
| Thermometer | Glass / Digital | Measures the temperature of the water accurately |
| Lid | Insulated | Prevents heat escape from the top |
Fig.: A calorimeter works just like a thermos flask — both trap heat inside using insulation
The Principle of Calorimetry
The entire working of a calorimeter is based on one fundamental law of physics — the Law of Conservation of Energy. Since the calorimeter is thermally insulated, no heat enters or leaves the system. This means:
Principle of Calorimetry: In an isolated system, the total heat lost by the hot body is exactly equal to the total heat gained by the cold body.
Heat Lost = Heat Gained
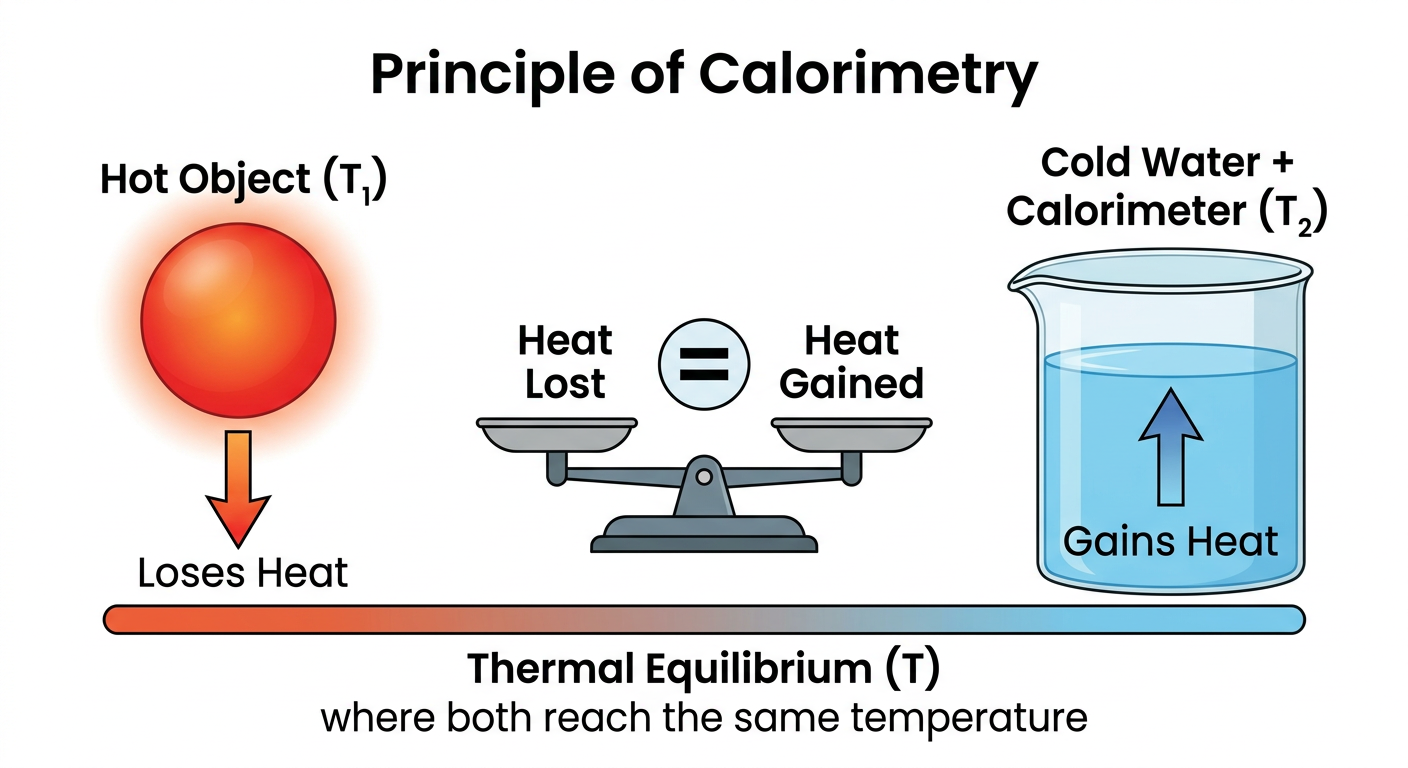
Fig.: Heat flows from the hot object to the cold water and calorimeter until thermal equilibrium is reached
Experiment: Method of Mixtures
The method of mixtures is the standard experimental technique for determining the specific heat capacity of an unknown substance using a calorimeter.
Procedure:
- Fill the Calorimeter: Pour a known mass of water (m₃) into the inner copper vessel. Note the initial temperature (T₂) of both water and calorimeter.
- Heat the Sample: Heat the sample (substance whose specific heat you want to find) to a high temperature (T₁). For example, place it in boiling water at 100°C.
- Quick Transfer: Quickly transfer the hot sample into the calorimeter water. Speed matters — minimise heat loss during the transfer!
- Stir Continuously: Use the stirrer to ensure uniform temperature distribution throughout the water.
- Record Final Temperature: Wait until the thermometer shows a stable reading. This is the final equilibrium temperature (T).
- Apply the Formula: Use the calorimetry equation: Heat lost by sample = Heat gained by water + Heat gained by calorimeter.
Derivation of Calorimetry Equation
Let's build the master formula step by step. First, define all our variables clearly.
Variable Definitions
| Symbol | Meaning |
|---|---|
| m₁ | Mass of the hot sample |
| m₂ | Mass of the calorimeter (+ stirrer) |
| m₃ | Mass of water in the calorimeter |
| s₁ | Specific heat capacity of the sample (unknown) |
| s₂ | Specific heat capacity of calorimeter material (e.g., copper = 387 J kg⁻¹ K⁻¹) |
| s₃ | Specific heat capacity of water = 4186 J kg⁻¹ K⁻¹ |
| T₁ | Initial temperature of the hot sample |
| T₂ | Initial temperature of calorimeter + water |
| T | Final equilibrium temperature of the entire system |
Step-by-Step Derivation
The sample cools from T₁ down to T, so ΔT = T₁ − T
The calorimeter heats up from T₂ to T, so ΔT = T − T₂
Water also heats from T₂ to T (same ΔT as the calorimeter)
m₁ s₁ (T₁ − T) = m₂ s₂ (T − T₂) + m₃ s₃ (T − T₂) ... Eq. (1)
This is the master formula. If you know all masses, temperatures, and specific heats of water and copper, you can calculate s₁ for any unknown material.
Example
Problem: A 0.06 kg aluminium sphere is heated to 100°C and placed in a 0.12 kg copper calorimeter containing 0.30 kg of water at 25°C. The mixture reaches a steady temperature of 28°C. Find the specific heat capacity of aluminium.
Solution:
Given:
Mass of aluminium sphere (m₁) = 0.06 kg
Mass of copper calorimeter (m₂) = 0.12 kg
Mass of water (m₃) = 0.30 kg
Specific heat of copper (s₂) = 387 J kg⁻¹ K⁻¹
Specific heat of water (s₃) = 4180 J kg⁻¹ K⁻¹
Initial temp of sphere (T₁) = 100°C
Initial temp of water + calorimeter (T₂) = 25°C
Final equilibrium temp (T) = 28°C
Step 1: Calculate temperature changes
- Temperature drop of sphere: T₁ − T = 100 − 28 = 72°C
- Temperature rise of water & calorimeter: T − T₂ = 28 − 25 = 3°C
Step 2: Apply Eq.
Step 3: Substitute values
Step 4: Compute the numerator
- 0.12 × 387 = 46.44
- 0.30 × 4180 = 1254
- Sum = 46.44 + 1254 = 1300.44
- Numerator = 1300.44 × 3 = 3901.32
Step 5: Compute the denominator
- 0.06 × 72 = 4.32
Step 6: Final answer
Key Points: Calorimetry
- Calorimeter: An insulated device used to measure heat transfer during a process.
- Principle of Calorimetry: Heat lost = Heat gained (energy is conserved).
- Method of Mixtures: Heat a sample, place it in the calorimeter, and measure the temperature change to calculate the heat.
- Formula: Used to calculate the specific heat of a substance from measured temperatures and masses.
- Good Practice: Transfer the sample quickly and stir well for accurate results.
- Why Water is Used: Water has high specific heat, so it absorbs a lot of heat without a large temperature change.
