Advertisements
Advertisements
प्रश्न
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
उत्तर
The order of the reaction = `1/2+2`
= `2 1/2`
= 2.5
APPEARS IN
संबंधित प्रश्न
Define “zero order reaction”.
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
What is pseudo first order reaction? Give one· example of it.
For the first order thermal decomposition reaction, the following data were obtained:
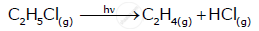
Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
Write two factors that affect the rate of reaction.
For a reaction A + B ⟶ P, the rate is given by
Rate = k [A] [B]2
What is the overall order of reaction if A is present in large excess?
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
Define the following terms:
Half-life period of reaction (t1/2).
Which of the following statements is not correct about order of a reaction.
The value of rate constant of a pseudo first order reaction ______.
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
At concentration of 0.1 and 0.2 mol L–1 the rates of deem position of a compound were found to be 0.18 and 0.72 mol L–1 m–1. What is the order of the reaction?
On heating compound (A) gives a gas (B) which is constituent of air. The gas when treated with H2 in the presence of catalyst gives another gas (C) which is basic in nature, (A) should not be ______.
Which of the following statement is true?
