Topics
Gravitation
- Concept of Gravitation
- Force
- Motion and Rest
- Centripetal Acceleration and Centripetal Force
- Kepler’s Laws
- Newton’s Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational force
- Acceleration Due to Gravity (Earth’s Gravitational Acceleration)
- Concept of Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Weightlessness in Space
Periodic Classification of Elements
- History of Periodic Table: Early Attempts at the Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Merits and Demerits of Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Properties
- Valency
- Atomic Radius Or Atomic Size
- Metallic and Non-metallic Characters
- Group VIIA Or Group 17 (The Halogens)
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equation
- Balancing Chemical Equation
- Types of Chemical Change or Chemical Reaction
- Direct Combination (or Synthesis) Reaction
- Decomposition Reactions
- Single Displacement Reactions
- Double Displacement Reaction
- Energy Change in Chemical Reactions
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Oxidation, Reduction and Redox Reactions
- Corrosion of Metals
- Rancidity of Food and Its Prevention
Effects of Electric Current
- Electric Circuit
- Ohm's Law (V = IR)
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Magnetic Field Due to Current in a Loop (Or Circular Coil)
- Magnetic Field Due to a Current Carving Cylindrical Coil (or Solenoid)
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Fleming’s Right Hand Rule
- Types of Current
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Spherical Lens
- Convex Lens
- Images Formed by Convex Lenses
- Concave Lens
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification Due to Spherical Lenses
- Power of a Lens
- Combination of Lenses
- Human Eye
- Working of the Human Eye
- Eye Defect and Its Correction: Myopia Or Near-sightedness
- Eye Defect and its Correction: Hypermetropia or Far-sightedness
- Eye Defect and Its Correction: Presbyopia
- Persistence of Vision
Metallurgy
- Types of Element: Metals
- Physical Properties of Metals
- Chemical Properties of Metal
- Reactions of Metal
- Reactivity Series of Metals
- Types of Element: Non-metal
- Physical Properties of Non-metal
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy
- Extraction of Reactive Metals
- Extraction of Aluminium
- Extraction of Moderately Reactive Metals
- Extraction of Less Reactive Metals
- Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Carbon Compounds in Everyday Life
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Properties of Carbon
- Hydrocarbons
- Structural Variations of Carbon Chains in Hydrocarbons
- Functional Groups in Carbon Compounds
- Homologous Series of Carbon Compound
- Nomenclature of Organic Compounds
- The IUPAC System of Nomenclature
- Chemical Properties of Carbon Compounds
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
- Concept of Space Missions
- Artificial Satellites
- Types of Satellite
- Orbits of Artificial Satellites
- Space Launch Technology
- Space Missions Away from Earth
- India’s Space Programmes: Chandrayaan – 1
- India’s Space Programmes: Chandrayaan – 2
- India’s Space Programmes: Chandrayaan – 3
- India’s Space Programmes: Mangalyaan (Mars vehicle)
- India’s Space Programmes: Missions to Other Planets
- India and Space Technology
- Space Debris and Its Management
School of Elements
The Magic of Chemical Reactions
The Acid Base Chemistry
- Properties of Acids
- Strength of Acidic or Basic Solutions
- Strength of Acidic or Basic Solutions
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic Force
- The Bar Magnet
- Right-hand Thumb Rule
- Magnetic Field Due to Current in a Loop (Or Circular Coil)
- Magnetic Field Due to a Current Carving Cylindrical Coil (or Solenoid)
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- Alternating Current (A.C.) Generator
- Direct Current Motor
- Household Electrical Circuits
Wonders of Light 1
- Spherical Mirrors
- Concave Mirror
- Concave Mirror
- Sign Convention
- Linear Magnification (M) Due to Spherical Mirrors
- Images Formed by Sperical Lenses
- Convex Lens
- Sign Convention
- Magnification Due to Spherical Lenses
- Power of a Lens
- Human Eye
- Eye Defect and Its Correction: Myopia Or Near-sightedness
- Spherical Mirrors
Wonders of Light 2
Striving for better Environment 1
- Pollution and Its Types
- Air Pollution and Its Causes
- Effects of Air Pollution
- Water Pollution and Its Causes
- Effects of Water Pollution
- Soil Pollution and its Causes
- Effects of Soil Pollution
- Noise Pollution
- Radioactive Pollution and Effects
- Abatement of Pollution
- Sustainable Use of Resources
- IUPAC Nomenclature: Structure and Components
- Steps for IUPAC Nomenclature
IUPAC Nomenclature: Structure and Components
IUPAC stands for the International Union of Pure and Applied Chemistry. It was established in 1919 to replace the International Congress of Applied Chemistry. The headquarters of this organisation is in the United States. It is a standardised system for naming chemical compounds. This system provides unique and universally accepted names based on the molecular structure of compounds.
IUPAC names consist of three main components: Prefix - Parent - Suffix
Components of the IUPAC Naming System:
| Component | Description | Example |
|---|---|---|
| Root (Parent) | The longest continuous carbon chain in the molecule. | "Meth-" (1C), "Eth-" (2C), "Prop-" (3C) |
| Suffix | Indicates the main functional group in the molecule. | "-ol" (Alcohol), "-al" (Aldehyde), "-oic acid" (Carboxylic Acid) |
| Prefix | Denotes substituents or side chains attached to the parent chain. | "Chloro-", "Bromo-", "Methyl-" |
Steps for IUPAC Nomenclature
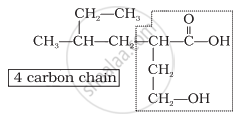
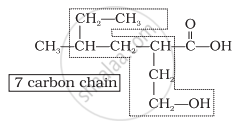
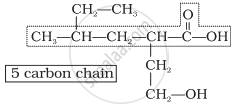
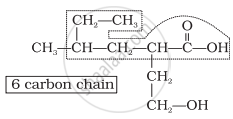
Step 1: Identify the Parent Chain
- Count the number of carbon atoms in the longest chain.
- Assign the corresponding alkane base name:
| Number of Carbons | Root Name |
|---|---|
| 1 | Meth- |
| 2 | Eth- |
| 3 | Prop- |
| 4 | But- |
| 5 | Pent- |
| 6 | Hex- |
| 7 | Hept- |
| 8 | Oct- |
| 9 | Non- |
| 10 | Dec- |
Step 2: Identify the Functional Group
- Replace the last ‘e’ from the alkane name with the functional group suffix.
- If the functional group is a halogen, it is added as a prefix.
| Functional Group | Structure | IUPAC Suffix |
|---|---|---|
| Alkene | C=C | -ene |
| Alkyne | C≡C | -yne |
| Alcohol | -OH | -ol |
| Aldehyde | -CHO | -al |
| Ketone | C=O | -one |
| Nitrile | C≡N | -nitrile |
| Carboxylic Acid | -COOH | -oic acid |
| Carboxylate Ion | -COO⁻ | -oate |
| Ester | -COOR | -oate |
| Amine | -NH₂ | -amine |
| Amide | -CONH₂ | -amide |
Step 3: Assign Numbering to the Carbon Chain
- Number the chain from the end nearest to the functional group.
- The functional group must get the lowest possible number.
- If multiple functional groups are present, assign the lowest number to the highest-priority group.
- For multiple identical groups, use prefixes like di-, tri-, tetra- (e.g., 2,3-dimethylpentane).
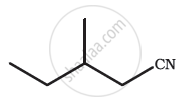
| Sr. No. | Structural Formula | Two Numberings of the Carbon Chain | Acceptable Numbering | IUPAC Name of the Compound |
| 1 | \[\begin{array}{cc} \phantom{}\ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{OH}\phantom{}\\ \end{array}\] |
\[\begin{array}{cc} \phantom{.}\ce{C^{1} - C^{2} - C^{3}}\\ \phantom{}|\\ \phantom{..}\ce{OH}\\ \phantom{.}\ce{C^{3} - C^{2} - C^{1}}\\ \phantom{}|\\ \phantom{..}\ce{OH} \end{array}\] |
Both numberings equivalent | propan-2-ol |
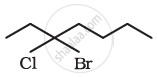
| 2 | \[\begin{array}{cc} \phantom{}\ce{CH3 - CH2 - CH2 - CH - CH3}\\ \phantom{................}|\phantom{...}\\ \phantom{..............}\ce{Cl}\phantom{}\\ \end{array}\] |
\[\begin{array}{cc} \phantom{.}\ce{C^{1} - C^{2} - C^{3} - C^{4} - C^{5}}\\ \phantom{...........}|\\ \phantom{...........}\ce{Cl}\\ \phantom{.}\ce{C^{5} - C^{4} - C^{3} - C^{2} - C^{1}}\\ \phantom{...........}|\\ \phantom{...........}\ce{Cl}\\ \end{array}\] |
\[\begin{array}{cc} \phantom{.}\ce{C^{5} - C^{4} - C^{3} - C^{2} - C^{1}}\\ \phantom{}|\\ \phantom{.}\ce{Cl}\\ \end{array}\] |
2-chloropentane |
| 3 | \[\begin{array}{cc} \ce{O}\phantom{...............}\\ ||\phantom{...............}\\ \phantom{}\ce{CH3 - C - CH2 - CH2 - CH3}\\ \end{array}\] |
\[\begin{array}{cc} \ce{O}\phantom{............}\\ ||\phantom{............}\\ \phantom{}\ce{C1 - C2 - C3 - C4 - C5}\\ \end{array}\] \[\begin{array}{cc} \ce{O}\phantom{............}\\ ||\phantom{............}\\ \phantom{}\ce{C5 - C4 - C3 - C2 - C1}\\ \end{array}\] |
\[\begin{array}{cc} \ce{O}\phantom{............}\\ ||\phantom{............}\\ \phantom{}\ce{C1 - C2 - C3 - C4 - C5}\\ \end{array}\] |
pentan-2-one |
Examples of IUPAC Naming
| Sr. No. | Common Name | Structural Formula | IUPAC Name |
|---|---|---|---|
| 1 | Ethylene | CH₂=CH₂ | Ethene |
| 2 | Acetylene | HC≡CH | Ethyne |
| 3 | Acetic acid | CH₃-COOH | Ethanoic acid |
| 4 | Methyl alcohol | CH₃-OH | Methanol |
| 5 | Ethyl alcohol | CH₃-CH₂-OH | Ethanol |
| 6 | Acetaldehyde | CH₃-CHO | Ethanal |
| 7 | Acetone | CH₃-CO-CH₃ | Propanone |
| 8 | Ethyl methyl ketone | CH₃-CO-CH₂-CH₃ | Butan-2-one |
| 9 | Ethyl amine | CH₃-CH₂-NH₂ | Ethanamine |
| 10 | n-Propyl chloride | CH₃-CH₂-CH₂-Cl | 1-Chloropropane |
If you would like to contribute notes or other learning material, please submit them using the button below.