Topics
Gravitation
- Concept of Gravitation
- Force
- Motion and Rest
- Centripetal Acceleration and Centripetal Force
- Kepler’s Laws
- Newton’s Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational force
- Acceleration Due to Gravity (Earth’s Gravitational Acceleration)
- Concept of Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Weightlessness in Space
Periodic Classification of Elements
- History of Periodic Table: Early Attempts at the Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Merits and Demerits of Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Properties
- Valency
- Atomic Radius Or Atomic Size
- Metallic and Non-metallic Characters
- Group VIIA Or Group 17 (The Halogens)
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equation
- Balancing Chemical Equation
- Types of Chemical Change or Chemical Reaction
- Direct Combination (or Synthesis) Reaction
- Decomposition Reactions
- Single Displacement Reactions
- Double Displacement Reaction
- Energy Change in Chemical Reactions
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Oxidation, Reduction and Redox Reactions
- Corrosion of Metals
- Rancidity of Food and Its Prevention
Effects of Electric Current
- Electric Circuit
- Ohm's Law (V = IR)
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Magnetic Field Due to Current in a Loop (Or Circular Coil)
- Magnetic Field Due to a Current Carving Cylindrical Coil (or Solenoid)
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Fleming’s Right Hand Rule
- Types of Current
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Spherical Lens
- Convex Lens
- Images Formed by Convex Lenses
- Concave Lens
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification Due to Spherical Lenses
- Power of a Lens
- Combination of Lenses
- Human Eye
- Working of the Human Eye
- Eye Defect and Its Correction: Myopia Or Near-sightedness
- Eye Defect and its Correction: Hypermetropia or Far-sightedness
- Eye Defect and Its Correction: Presbyopia
- Persistence of Vision
Metallurgy
- Types of Element: Metals
- Physical Properties of Metals
- Chemical Properties of Metal
- Reactions of Metal
- Reactivity Series of Metals
- Types of Element: Non-metal
- Physical Properties of Non-metal
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy
- Extraction of Reactive Metals
- Extraction of Aluminium
- Extraction of Moderately Reactive Metals
- Extraction of Less Reactive Metals
- Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Carbon Compounds in Everyday Life
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Properties of Carbon
- Hydrocarbons
- Structural Variations of Carbon Chains in Hydrocarbons
- Functional Groups in Carbon Compounds
- Homologous Series of Carbon Compound
- Nomenclature of Organic Compounds
- The IUPAC System of Nomenclature
- Chemical Properties of Carbon Compounds
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
- Concept of Space Missions
- Artificial Satellites
- Types of Satellite
- Orbits of Artificial Satellites
- Space Launch Technology
- Space Missions Away from Earth
- India’s Space Programmes: Chandrayaan – 1
- India’s Space Programmes: Chandrayaan – 2
- India’s Space Programmes: Chandrayaan – 3
- India’s Space Programmes: Mangalyaan (Mars vehicle)
- India’s Space Programmes: Missions to Other Planets
- India and Space Technology
- Space Debris and Its Management
School of Elements
The Magic of Chemical Reactions
The Acid Base Chemistry
- Properties of Acids
- Strength of Acidic or Basic Solutions
- Strength of Acidic or Basic Solutions
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic Force
- The Bar Magnet
- Right-hand Thumb Rule
- Magnetic Field Due to Current in a Loop (Or Circular Coil)
- Magnetic Field Due to a Current Carving Cylindrical Coil (or Solenoid)
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- Alternating Current (A.C.) Generator
- Direct Current Motor
- Household Electrical Circuits
Wonders of Light 1
- Spherical Mirrors
- Concave Mirror
- Concave Mirror
- Sign Convention
- Linear Magnification (M) Due to Spherical Mirrors
- Images Formed by Sperical Lenses
- Convex Lens
- Sign Convention
- Magnification Due to Spherical Lenses
- Power of a Lens
- Human Eye
- Eye Defect and Its Correction: Myopia Or Near-sightedness
- Spherical Mirrors
Wonders of Light 2
Striving for better Environment 1
- Pollution and Its Types
- Air Pollution and Its Causes
- Effects of Air Pollution
- Water Pollution and Its Causes
- Effects of Water Pollution
- Soil Pollution and its Causes
- Effects of Soil Pollution
- Noise Pollution
- Radioactive Pollution and Effects
- Abatement of Pollution
- Sustainable Use of Resources
- Reaction of Metals with Oxygen
- Reaction of Metals with Water
- Experiment 1
- Reaction of Metals with Acids
- Experiment 2
- Reaction of Metals with Nitric Acid
- Reaction of Metals with Salts of Other Metals
- Experiment 3
- Reaction of Metals with Nonmetals
Reaction of Metals with Oxygen
Metals react with oxygen to form metal oxides. The rate of reaction depends on the metal’s reactivity. Highly reactive metals, such as sodium and potassium, react at room temperature, while others, like magnesium, require heating.
1. Highly reactive metals (e.g., sodium, potassium) react at room temperature:
4Na(s) + O₂(g) → 2Na₂O(s)
Sodium is stored in kerosene to prevent accidental combustion.
2. Moderately reactive metals (e.g., magnesium) react upon heating:
Na₂O(s) + H₂O(l) → 2NaOH(aq)
Magnesium oxide reacts with water to form magnesium hydroxide:
2Mg(s) + O₂(g) → 2MgO(s)
Reaction of Metals with Water
The reaction of metals with water varies depending on their reactivity. Some metals react violently, some react slowly, and others do not react at all.
1. Highly reactive metals (e.g., sodium, potassium) react explosively, producing hydrogen gas and heat:
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g) + heat
2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g) + heat
2. Moderately reactive metals (e.g., calcium) react slowly, producing hydrogen gas in the form of bubbles:
2Ca(s) + 2H₂O(l) → 2Ca(OH)₂(aq) + H₂(g)
3. Less reactive metals (e.g., aluminium, zinc, iron) do not react with cold or hot water but react with steam:
2Al(s) + 3H₂O(g) → Al₂O₃(s) + 3H₂(g)
3Fe(s) + 4H₂O(g) → Fe₃O₄(s) + 4H₂(g)
Experiment 1
1. Aim: To observe how different metals react with water.
2. Requirements: Beakers, water, and metal samples (excluding sodium for safety).
3. Procedure
- Drop a small piece of each metal into separate beakers filled with water.
- Observe whether the metal reacts, floats, or sinks.
- Record reaction rate, gas formation, and heat generation.
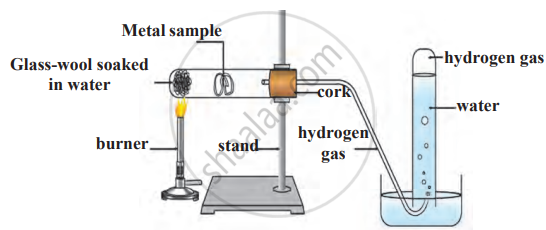
Reaction of a metal with water
4. Observations
- Sodium and potassium react violently.
- Calcium reacts slowly, forming bubbles.
- Zinc, iron, and aluminium react only with steam.
5. Conclusion: Metals have different reactivity levels with water. Highly reactive metals produce hydrogen gas and form alkalis, while less reactive metals react only with steam.
Reaction of Metals with Acids
Most metals react with dilute acids to form salt and hydrogen gas. The reactivity of metals varies, with some reacting vigorously and others reacting slowly.
Reactivity order: Mg > Al > Zn > Fe
1. Highly reactive metals (e.g., magnesium) react vigorously:
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
2. Moderately reactive metals (e.g., aluminium, zinc, iron) react at different speeds:
2Al(s)+6HCl(aq)→2AlCl₃(aq)+3H₂(g)
Fe(s) + 2HCl(aq) → FeCl₂(aq) + H₂(g)
Zn(s) + HCl(aq) → ZnCl₂(aq) + H₂(g)
The hydrogen gas released can be tested by bringing a burning splint near it, which produces a ‘pop’ sound.
Experiment 2
1. Aim: To study the reaction of metals with dilute acids.
2. Requirements: Test tubes, dilute hydrochloric acid, and samples of magnesium, aluminium, zinc, and iron.
3. Procedure
- Add a small piece of each metal to separate test tubes containing dilute hydrochloric acid.
- Observe gas bubble formation.
- Test the gas with a burning splint.
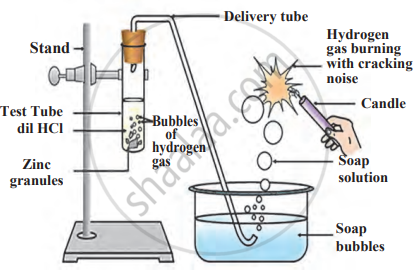
Reaction of metals with dilute acid
4. Observations
- Magnesium reacts vigorously, producing hydrogen gas.
- Aluminium, zinc, and iron react at different speeds.
- The gas burns with a ‘pop’ sound.
5. Conclusion: Metals react with acids to form salts and hydrogen gas. The reaction speed varies with different metals.
Reaction of Metals with Nitric Acid
Metals react with nitric acid to form metal nitrates and release nitrogen oxides (NO, NO₂, or N₂O) instead of hydrogen gas. This is because nitric acid is a strong oxidising agent, which oxidises the hydrogen gas into water. However, magnesium and manganese react with very dilute nitric acid to produce hydrogen gas.
1. Reaction with concentrated nitric acid
Copper reacts with concentrated nitric acid to form copper nitrate and nitrogen dioxide gas:
Cu(s) + 4HNO₃(aq) → Cu(NO₃)₂(aq) + 2NO₂(g) + 2H₂O(l)
NO gas is colourless but turns brown in air due to oxidation into NO₂.
2. Reaction with dilute nitric acid
Copper reacts with dilute nitric acid to form copper nitrate and nitric oxide (NO) gas:
3Cu(s) + 8HNO₃(aq) → 3Cu(NO₃)₂(aq) + 2NO(g) + 4H₂O(l)
Aqua Regia: It is a highly corrosive mixture of concentrated hydrochloric acid (HCl) and nitric acid (HNO₃) in a 3:1 ratio. It dissolves noble metals like gold and platinum, which do not react with nitric acid alone.
Au(s) + HNO₃ + HCl → H[AuCl₄] + NO
Reaction of Metals with Salts of Other Metals
A more reactive metal can displace a less reactive metal from its salt solution. This is known as a displacement reaction.
Iron displaces copper from copper sulphate solution:
Fe(s) + CuSO₄(aq) → FeSO₄(aq) + Cu(s)
The iron nail gets coated with copper, showing that iron is more reactive than copper.
Experiment 3
1. Aim: To observe displacement reactions between metals and metal salts.
2. Requirements: Test tubes, iron nails, copper wire, solutions of copper sulphate and ferrous sulphate.
3. Procedure
- Place an iron nail in copper sulphate solution.
- Place a copper wire in ferrous sulphate solution.
- Observe changes over time.

Reaction of metal with solution of salts of other metals
4. Observations
- The iron nail develops a copper coating, indicating a reaction.
- No reaction is seen in the copper wire with ferrous sulphate solution.
5. Conclusion: A more reactive metal can replace a less reactive metal from its salt solution, confirming the reactivity series.
Reaction of Metals with Nonmetals
Metals react with nonmetals to form ionic compounds. This reaction occurs because metals lose electrons to achieve a stable electron configuration, while nonmetals gain electrons to complete their outermost shell. Noble gases (like helium, neon, and argon) do not react because they already have a complete octet, making them chemically inert.
When a metal reacts with a nonmetal, an electron transfer takes place, forming an ionic bond. The metal donates electrons, becoming a cation (+), while the nonmetal accepts electrons, becoming an anion (-).
Example,
1. Formation of Sodium Chloride (NaCl)
2Na(s) + Cl₂(g) → 2NaCl(s)
- Sodium (Na) loses one electron to form Na⁺.
- Chlorine (Cl₂) gains electrons to form Cl⁻.
- The oppositely charged ions attract, forming an ionic bond in NaCl.
Similarly, magnesium and potassium also form ionic compounds with chlorine:
2. Formation of Magnesium Chloride (MgCl₂)
Mg(s) + Cl₂(g) → MgCl₂(s)
- Magnesium loses two electrons to form Mg²⁺.
- Each chlorine atom gains one electron, forming Cl⁻ ions.
3. Formation of Potassium Chloride (KCl)
K(s) + Cl₂(g) → 2KCl(s)
- Potassium loses one electron to form K⁺.
- Chlorine gains an electron to form Cl⁻.
