Advertisements
Advertisements
प्रश्न
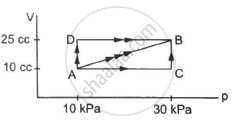
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
उत्तर
Work done during any process, W = P ∆ V
If both pressure and volume are changing during a process, then work done can be found out by finding the area under the PV diagram.
In path ACB, for line AC :-
Since initial volume is equal to final volume,
∆ V = 0
⇒ WAC = P ∆ V = 0
For line BC :-
P = 30 × 103 pa
WACB = WAC + WBC = 0 + P∆V
= 30 × 103 × (25 − 10) × 10−6
= 0.45 J
For path AB:-
Since both pressure and volume are changing, we use the mean pressure to find the work done.
Mean pressure, P = \[\frac{1}{2} \times (30 + 10) \times {10}^3\]
WAB = \[\frac{1}{2}\]× (10 + 30) × 103 × 15 × 10−6
= \[\frac{1}{2}\] × 40 × 15 × 10−3 = 0.30 J
Initial volume in path ADB, along line DB is the same as final volume. Thus, work done along this line is zero.
Along line AD, P = 10 kPa
W = WAD + WDB
= 10 × 103 (25 − 10) × 10−6 + 0
= 10 × 15 × 10−3 = 0.15 J
APPEARS IN
संबंधित प्रश्न
Should the internal energy of a system necessarily increase if heat is added to it?
When we rub our hands they become warm. Have we supplied heat to the hands?
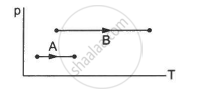
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
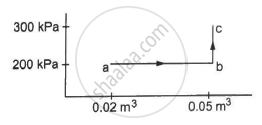
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.
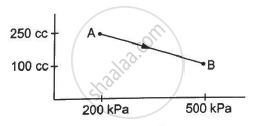
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.
A mixture of fuel and oxygen is burned in a constant-volume chamber surrounded by a water bath. It was noticed that the temperature of water is increased during the process. Treating the mixture of fuel and oxygen as the system,
- Has heat been transferred?
- Has work been done?
- What is the sign of ∆U?
A system releases 130 kJ of heat while 109 kJ of work is done on the system. Calculate the change in internal energy.
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
A thermodynamic system goes from states
(i) P, V to 2P, V (ii) P, V to P, 2V
The work done in the two cases is ____________.
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in internal energy of the gas?
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in the temperature of the gas?
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
n mole of a perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes:
A `→` B: Isothermal expansion at temperature T so that the volume is doubled from V1 to V2 = 2V1 and pressure changes from P1 to P2.
B `→` C: Isobaric compression at pressure P2 to initial volume V1.
C `→` A: Isochoric change leading to change of pressure from P2 to P1.
Total workdone in the complete cycle ABCA is ______.

The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
