Advertisements
Advertisements
Question
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is doubled?
Solution
Rate of reaction, R = k [A]2
If the concentration of the reactant is doubled, i.e. [A] = 2R, then the rate of the reaction would be R = k(2R)2
= 4kR2
= 4 R
Therefore, the rate of the reaction would increase by 4 times.
APPEARS IN
RELATED QUESTIONS
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a reaction A + B ⟶ P, the rate is given by
Rate = k [A] [B]2
How is the rate of reaction affected if the concentration of B is doubled?
For a reaction : 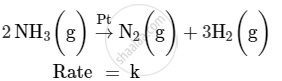
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
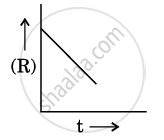
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume :
SO2Cl2 (g) → SO2 (g) + Cl2 (g)
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
Calculate the rate constant.
(Given : log 4 = 0.6021, log 2 = 0.3010)
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
Mention the factors that affect the rate of a chemical reaction.
In a reaction between A and B, the initial rate of reaction (r0) was measured for different initial concentrations of A and B as given below:
| A/mol L−1 | 0.20 | 0.20 | 0.40 |
| B/mol L−1 | 0.30 | 0.10 | 0.05 |
| r0/mol L−1 s−1 | 5.07 × 10−5 | 5.07 × 10−5 | 1.43 × 10−4 |
What is the order of the reaction with respect to A and B?
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
Define the following terms:
Half-life period of reaction (t1/2).
Rate of reaction for the combustion of propane is equal to:
\[\ce{C3H8_{(g)} + 5O2_{(g)} -> 3CO2_{(g)} + 4H2O_{(g)}}\]
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?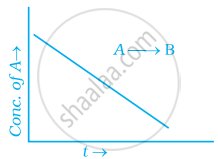
Why is the probability of reaction with molecularity higher than three very rare?
Why does the rate of any reaction generally decreases during the course of the reaction?
Why can we not determine the order of a reaction by taking into consideration the balanced chemical equation?
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
