Topics
Matter
- Matter (Substance)
- Characteristics of Particles (Molecules) of Matter
- States of Matter
- The Solid State
- The Liquid State
- The Gaseous State
- Kinetic Theory of Matter
- Heat and change of physical state
- Concept of Melting (Fusion)
- Concept of Evaporation
- Concept of Boiling (Vaporization)
- Concept of Condensation (Liquefaction)
- Concept of Freezing (Solidification)
- Concept of Sublimation
- Concept of Desublimation (Deposition)
- Laws of Chemical Combination
- Law of Conservation of Mass
Physical and Chemical Changes
- Changes-Physical and Chemical
- Classification of Change: Slow and Fast Changes
- Classification of Change: Natural and Man-made Changes
- Classification of Change: Periodic and Non-periodic Changes
- Classification of Change: Reversible and Irreversible Changes
- Classification of Change: Physical Changes
- Chemical Reaction
Elements, Compounds and Mixtures
- Matter (Substance)
- Natural substances
- Pure Substances
- Impure Substance
- Elements
- Types of Element: Metals
- Types of Element: Non-metal
- Type of Element: Metalloid
- Type of Elements: Noble Or Inert Gases
- Difference Between Metals, Non-metals, Metalloids, and Noble Gases
- Compound
- Mixture
- Types of Mixtures
- Formation of Mixtures
- Separation of Mixtures
- Methods of Separation
- Handpicking Method
- Magnetic Separation Method
- Gravitation Method
- Sublimation Method
- Solvent Extraction (Using a Separating Funnel Method)
- Crystallisation Method
- Sedimentation Method
- Decantation Method
- Filtration Method
- Evaporation Method
- Centrifugation Method
- Fractional Distillation Method
- Chromatography Method
- Diffusion Method
- Liquefaction Method
Atomic Structure
- Atoms: Building Blocks of Matter
- History of Atom
- Dalton’s Atomic Theory
- J. J. Thomson’s Atomic Model
- Lord Rutherford’s Atomic model
- Discovery of Charged Particles in Matter
- Electrons (e)
- Protons (p)
- Nucleus
- Neutrons (n)
- Neils Bohr’s Model of an Atom
- Structure of an Atom
- Atomic Number (Z), Mass Number (A), and Number of Neutrons (n)
- Atomic Mass
- Isotopes
- Electronic Configuration of Atom
- Electronic Configuration of Atom
- Valency
- Variable Valency
- Ions (Radicals) and Its Types
Language of Chemistry
- Language of Chemistry
- Law of Conservation of Mass
- Symbols Used to Represent Atoms of Different Elements
- Valency
- Variable Valency
- Ions (Radicals) and Its Types
- Chemical Formula or Molecular Formula
- Molecular Formula of Compounds
- Chemical Formula or Molecular Formula
- Significance of Molecular/Chemical Formulae
- Chemical Equation
- Balancing Chemical Equation
Chemical Reactions
Chemical Reaction
- Chemical Reaction
- Chemical Reaction
- Indicators of a Chemical Change (Chemical Reaction)
- Conditions Necessary for Chemical Reactions
Types of Chemical Reactions
- Types of Chemical Change or Chemical Reaction
- Direct Combination (or Synthesis) Reaction
- Decomposition Reactions
- Single Displacement Reactions
- Double Displacement Reaction
- Types of Double Displacement: Precipitation Reaction
- Types of Double Displacement: Neutralization Reaction
- Neutralization Reactions in Our Daily Life
- Reactivity Series of Metals
- Energy Change in Chemical Reactions
- Oxides
Hydrogen
- Hydrogen
- Preparation of Hydrogen
- Laboratory Preparation of Hydrogen
- Manufacture of Hydrogen
- Physical Properties of Hydrogen
- Chemical Properties of Hydrogen
- Tests for Hydrogen
- Uses of Hydrogen
- Oxidation, Reduction and Redox Reactions
Water
- Water: Our Lifeline
- Importance of Water
- Sources of Water
- Water Cycle
- Availability of Water
- Physical Properties of Water
- Anomalous Expansion of Water
- Water - a Universal Solvent
- Capacity of Water to Dissolve Substances - Saturated, Unsaturated, and Supersaturated Solutions
- Solution
- Suspension Solution
- Colloidal Solution
- Difference Between Solution, Colloidal Solution, and Suspension Solution
- Crystals and Crystallisation
- Hydrated and Anhydrous Substances
- Efflorescence, Hygroscopic, and Deliquescence Substances
- Chemical Properties of Water
- Classification of water: Soft and Hard Water
- Advantage and Disadvantage of Hard Water
- Removal of Hardness of Water
- Water Pollution and Its Causes
- Prevention of Water Pollution
- Effects of Water Pollution
- Water Management (Conservation of Water)
- Fresh Water Management
- Waste Water Management
- Water - a Universal Solvent
Carbon and Its Compounds
- Carbon: A Versatile Element
- Classification of Compounds of Carbon
Allotropy in carbon
Amorphous Forms of Carbon
- Non-crystalline/Amorphous Forms: Coal
- Non-crystalline/Amorphous Forms: Coke
- Non-crystalline/Amorphous Forms: Charcoal
- Non-crystalline/Amorphous Forms: Lamp Black (Soot)
- Non-crystalline/Amorphous Forms: Gas Carbon
Carbon Dioxide
- Carbon Dioxide
- Preparation of Carbon Dioxide
- Laboratory Preparation of Carbon Dioxide
- Properties of Carbon Dioxide
- Tests for Carbon Dioxide
- Importance of Carbon Dioxide
- Fire Extinguisher
- Carbon Dioxide
- Green House Effect
- Preventive Measures of Green House Effect
- Global Warming
- Preventive Measures of Global Warming
- Cause of Increased Percentage of Carbon Dioxide in the Atmosphere
- Steps to Balance Carbon Dioxide in the Atmosphere
Carbon Monoxide - A Compound of Carbon
- Carbon Monoxide
- Formation of Carbon Monoxide and Its Addition to the Atmosphere
- Harmful Effects of Carbon Monoxide
- Precautions and Remedies for Carbon Monoxide Poisoning
- Reducing Action of Carbon Monoxide
- Chemical and Physical Changes
- Experiment
Chemical and Physical Changes:
In everyday life, we come across both physical and chemical changes. A chemical change is when a substance transforms into a new material with a different composition and properties, while a physical change only affects the appearance or state of a substance without changing its chemical makeup.
- For example, the ripening of a mango or banana involves chemical reactions that change the fruit’s taste, texture, and colour. Enzymes convert starches into sugars, altering the fruit's composition and making it sweeter.
- In contrast, melting ice and boiling water are physical changes. Ice melting into water changes its state from solid to liquid, and boiling turns it into vapour, but the chemical structure of water (H₂O) stays the same.
- Similarly, dissolving salt in water is a physical process where the salt separates into ions but does not change chemically.

Some observations of chemical change
The image shows examples of various chemical changes.
- An apple turns brown, and bread becomes toasted, indicating a colour change.
- Nails rust over time, changing colour and forming rust.
- A matchstick burning demonstrates the release of energy as light and heat.
- Bananas become black as they ripen and decay, showing a colour and texture change.
- A colour change occurs in a chemical solution, indicating a reaction.
- Gas is released and bubbling is visible in a chemical reaction in a flask.
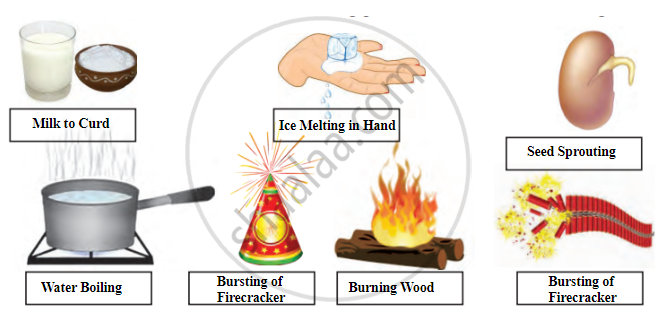
| Change | Description | Type of Change |
|---|---|---|
| Milk to Curd | Milk turns into curd. | Irreversible change (chemical) |
| Ice Melting in Hand | Ice melts into water. | Reversible change (physical) |
| Seed Sprouting | Seed sprouts and grows. | Irreversible change (biological/chemical) |
| Water Boiling | Water changes into steam. | Reversible change (physical) |
| Bursting of Firecracker | Firecrackers burst, producing sound, light, and smoke. | Irreversible change (chemical) |
| Burning Wood | Wood burns, turning into ash and smoke. | Irreversible change (chemical) |
- Fruit falling from a tree: physical change (movement).
- Rusting of iron: chemical change (new substance formed).
- Raining: physical change (change in state: water vapour to liquid).
- Lighting an electric bulb: physical change (no new substance formed, just energy transformation).
- Cutting vegetables: physical change (shape change, but still the same material).
Experiment
1. Aim: To observe and understand the characteristics of a chemical change by adding baking soda to lemon juice.
2. Requirements: lemon juice (in a clean glass), baking soda, a spoon, and clean apparatus for tasting.
3. Procedure
- Pour lemon juice into a clean glass.
- Take two drops of the lemon juice on a spoon and taste it. Note the sourness.
- Add a pinch of baking soda to the glass of lemon juice. Observe what happens:
- Do you see bubbles forming around the soda particles?
- Do you hear a faint fizzing sound when you put your ear close to the glass?
- Taste the mixture again. Compare the taste with the original lemon juice.
Observations
- Bubbles form, indicating the release of gas.
- A faint sound is heard due to the reaction.
- The baking soda particles dissolve in the liquid.
- The sour taste of the lemon juice becomes milder.
Conclusion: The change in taste and the formation of bubbles indicate a chemical change. A new substance with different properties is formed, showing that the composition of the original substances has changed.

