Advertisements
Advertisements
प्रश्न
Mention the factors that affect the rate of a chemical reaction.
उत्तर
The factors that affect the rate of a reaction are as follows:
(i) Nature of the reactant - The rate of reaction depends on the nature of the reactant. For instance, ionic molecules react more quickly than covalent ones.
(ii) State of reactants - Solid reactions are slower, liquid reactions are faster, and gas reactions are very quick.
(iii) Temperature - The rate of reaction is mainly affected by temperature. Every 10°C rise in temperature leads to an increased rate of reaction by 2-3 times.
`(r_(t + 10))/r_t` = 2 − 3
This ratio is called the temperature coefficient.
There are two reasons why the rate of reaction increases with increasing temperature.
- Increasing temperature raises the average kinetic energy of reactant molecules, increasing the rate of collisions.
- As temperature rises, the number of molecules with threshold energy increases, resulting in more active molecules. As a result, the number of effective collisions grows. As a result, the rate of reaction rises.
(iv) Concentration - The rate of reaction depends on the concentration of reactants.
Rate = k × Cn, where n = order of reaction, C = concentration of reactant.
(v) Catalyst - The presence of a catalyst changes the rate of the reaction. It lowers the activation energy by producing a chemical intermediate, lowering the potential energy barrier. Thus, the rate of reaction increases.
APPEARS IN
संबंधित प्रश्न
A → B is a first order reaction with rate 6.6 × 10-5m-s-1. When [A] is 0.6m, rate constant of the reaction is
- 1.1 × 10-5s-1
- 1.1 × 10-4s-1
- 9 × 10-5s-1
- 9 × 10-4s-1
A reaction is second order in A and first order in B.
(i) Write the differential rate equation.
(ii) How is the rate affected on increasing the concentration of A three times?
(iii) How is the rate affected when the concentrations of both A and B are doubled?
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For the first order thermal decomposition reaction, the following data were obtained:
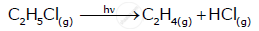
Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
Write two factors that affect the rate of reaction.
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
How does calcination differ from roasting?
Write the principle behind the following methods of refining:
Hydraulic washing
Which of the following statement is true for order of a reaction?
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be ______.
The value of rate constant of a pseudo first order reaction ______.
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
Why does the rate of any reaction generally decreases during the course of the reaction?
The rate constant for the reaction \[\ce{2H2O5 -> 4NO2 + O2}\] is 30 × 10–5 sec–1. if the rate is 204 × 10–5 mol L–1 S–1, then the concentration of N2O5 (in mol–1) is-
For a first order A → B, the reaction rate at reactant concentration of 0.01 m is found to be 2.0 × 10–5. The half-life period of reaction.
The conversion of molecules A to B follow second order kinetics. If concentration of A is increased to three times, how will it affect the rate of formation of B?
A drop of solution (volume 0.05 ml) contains 3.0 × 10-6 mole of H+. If the rate constant of disappearance of H+ is 1.0 × 107 mole l-1s-1. It would take for H+ in drop to disappear in ______ × 10-9s.
Assertion (A): Order of reaction is applicable to elementary as well as complex reactions.
Reason (R): For a complex reaction, molecularity has no meaning.
