Advertisements
Advertisements
प्रश्न
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
उत्तर
2-Methylbutane-1,3-diol
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Write the IUPAC name of the given compound:
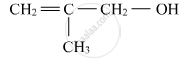
Name the following compound according to IUPAC system.
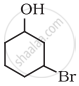
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{..........}\\
\ce{OH}\phantom{........}
\end{array}\]
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
\phantom{}|\phantom{....}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{.}\ce{Br}\phantom{...}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{.}}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
\phantom{}|\phantom{.............}|\phantom{......}|\phantom{.........}\\
\phantom{}\ce{OH}\phantom{..........}\ce{OH}\phantom{...}\ce{C2H5}\phantom{......}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
Write IUPAC name of the following compound:
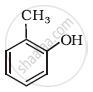
Write IUPAC name of the following compound:
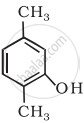
Write IUPAC name of the following compound:
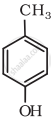
Write IUPAC name of the following compound:
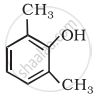
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{..........}|\\
\phantom{............}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
C6H5 – O – C2H5
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{...}|\\
\phantom{.....}\ce{CH3}
\end{array}\]
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give IUPAC name of the following ether:
CH3OCH2CH2Cl
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
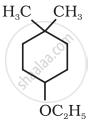
Give IUPAC name of the following ether:
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
How is phenol converted into the following?
benzoquinone
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Write the IUPAC name of the following compound:
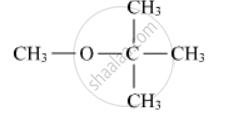
Write the IUPAC name of the following :
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Propanoic acid to ethylamine.
Resorcinol on distillation with zinc dust gives _________.
Write structural formulae for Methyl vinyl ether.
Write structural formulae for 1-Ethylcyclohexanol.
Write structural formulae for Cyclohex-2-en-1-ol.
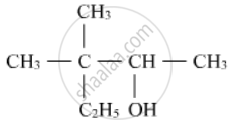
Write IUPAC name of the following
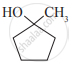
Write IUPAC names of the following
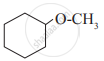
Glycerol is ____________.
Give IUPAC names of the following compound:
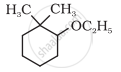
One of the following is not a dihydroxy derivative of benzene.
An example of a compound with functional group – O – is ____________.
Cresol has ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
HBr reacts fastest with ____________.
IUPAC name of m-cresol is ____________.
Ethylene reacts with Baeyer’s reagent to give ______.
When ethyl alcohol reacts with acetic acid, the products formed are:
Which of the following is most acidic?
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
The product of acid catalysed hydration of 2-phenylpropene is:
The heating of phenyl methyl ether with HI produces:
Which of the following gives a positive iodoform test?
Among the following sets of reactants which one produces anisole?
IUPAC name of m-cresol is ______.
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
What happens when benzene diazonium chloride is heated with water?
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Write steps to carry out the conversion of phenol to aspirin.
Explain why p-nitrophenol is more acidic than phenol.
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) | 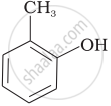 |
(a) Hydroquinone |
| (ii) | 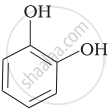 |
(b) Phenetole |
| (iii) | 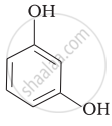 |
(c) Catechol |
| (iv) | 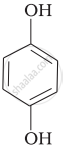 |
(d) o-Cresol |
| (v) | 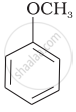 |
(e) guinone |
| (vi) | 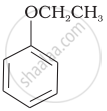 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Assertion: Phenols give o- and p-nitrophenol on nitration with conc. \[\ce{HNO3}\] and \[\ce{H2SO4}\] mixture.
Reason: –OH group in phenol is o–, p– directing.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
Identify A and B in the following:

Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Draw structure of the following compound.
2. 5-Diethylphenol
Draw structure of the following compound.
Prop-2-en-1-ol
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write structural formulae for:
p-Nitrophenol
The IUPAC name of 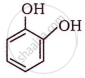 is ______.
is ______.
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
