Advertisements
Advertisements
Question
The work function of a photoelectric material is 4.0 eV. (a) What is the threshold wavelength? (b) Find the wavelength of light for which the stopping potential is 2.5 V.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Advertisements
Solution
Work function of a photoelectric material, ϕ = 4 eV = 4 × 1.6 × 10−19 J
Stopping potential, V0 = 2.5 V
Planck's constant, `h = 6.63 xx 10^-34 "Js"`
(a) Work function of a photoelectric material,
`phi = (hc)/λ_0`
Here, λ0 = threshold wavelength of light
c = speed of light
`therefore λ_0 = (hc)/phi`
`λ_0 = (6.63 xx 10^-34 xx 3 xx 10^8)/(4 xx 1.6 xx 10^-19)`
`λ_0 = (6.63 xx 3)/64 xx (10^27)/(10^-9)`
`λ_0 = 3.1 xx 10^-7 "m"`
`λ_0 = 310 "nm"`
(b) From Einstein's photoelectric equation,
`E = phi + eV_0`
On substituting the respective values , we get :-
`(hc)/λ = 4 xx 1.6 xx 10^-19 + 1.6 xx 10^-19 xx 2.5`
`⇒ λ = (6.63 xx 10^-34 xx 3 xx 10^8)/(6.5 xx 1.6 xx 10^-19)`
`⇒ λ = (6.63 xx 3 xx 10^-26)/(1.6 xx 10^-19 xx 6.5)`
`⇒ λ = 1.9125 xx 10^-7 = 191 "nm"`
APPEARS IN
RELATED QUESTIONS
Define the term 'intensity of radiation' in terms of photon picture of light.
Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
The work function for the following metals is given:
Na: 2.75 eV; K: 2.30 eV; Mo: 4.17 eV; Ni: 5.15 eV
Which of these metals will not give photoelectric emission for a radiation of wavelength 3300 Å from a He-Cd laser placed 1 m away from the photocell? What happens if the laser is brought nearer and placed 50 cm away?
Light of intensity 10−5 W m−2 falls on a sodium photo-cell of surface area 2 cm2. Assuming that the top 5 layers of sodium absorb the incident energy, estimate time required for photoelectric emission in the wave-picture of radiation. The work function for the metal is given to be about 2 eV. What is the implication of your answer?
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
Can we find the mass of a photon by the definition p = mv?
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
Can a photon be deflected by an electric field? Or by a magnetic field?
A hot body is placed in a closed room maintained at a lower temperature. Is the number of photons in the room increasing?
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
If the frequency of light in a photoelectric experiment is doubled, the stopping potential will ______.
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
When the sun is directly overhead, the surface of the earth receives 1.4 × 103 W m−2 of sunlight. Assume that the light is monochromatic with average wavelength 500 nm and that no light is absorbed in between the sun and the earth's surface. The distance between the sun and the earth is 1.5 × 1011 m. (a) Calculate the number of photons falling per second on each square metre of earth's surface directly below the sun. (b) How many photons are there in each cubic metre near the earth's surface at any instant? (c) How many photons does the sun emit per second?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A totally reflecting, small plane mirror placed horizontally faces a parallel beam of light, as shown in the figure. The mass of the mirror is 20 g. Assume that there is no absorption in the lens and that 30% of the light emitted by the source goes through the lens. Find the power of the source needed to support the weight of the mirror.
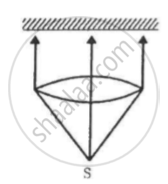
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, Show that the force on the sphere due to the light falling on it is the same even if the sphere is not perfectly absorbing.
Find the maximum kinetic energy of the photoelectrons ejected when light of wavelength 350 nm is incident on a cesium surface. Work function of cesium = 1.9 eV
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Find the maximum magnitude of the linear momentum of a photoelectron emitted when a wavelength of 400 nm falls on a metal with work function 2.5 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
- Assertion (A): For the radiation of a frequency greater than the threshold frequency, the photoelectric current is proportional to the intensity of the radiation.
- Reason (R): Greater the number of energy quanta available, the greater the number of electrons absorbing the energy quanta and the greater the number of electrons coming out of the metal.
