Advertisements
Advertisements
प्रश्न
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
उत्तर
Given rate = k[NO]2
Therefore, the order of the reaction = 2
Dimension of k = `"Rate"/["NO"]^2`
= `("mol L"^(-1) "s"^(-1))/("mol L"^(-1))^2`
= `("mol L"^(-1) "s"^(-1))/("mol"^2 "L"^(-2))`
= L mol−1 s−1
APPEARS IN
संबंधित प्रश्न
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
Write resonating structures of ozone.
Define the following terms:
Pseudo first-order reaction
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
In any unimolecular reaction:
(i) only one reacting species is involved in the rate determining step.
(ii) the order and the molecularity of slowest step are equal to one.
(iii) the molecularity of the reaction is one and order is zero.
(iv) both molecularity and order of the reaction are one.
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?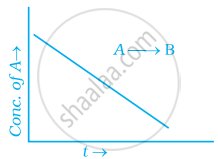
Why can’t molecularity of any reaction be equal to zero?
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Use Molecular Orbital theory to determine the bond order in each of species, [He2j+ and [He2]2+?
A catalyst in a reaction changes which of the following?
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
Identify the order of reaction from the following unit for its rate constant:
L mol–1s–1
The conversion of molecules A to B follow second order kinetics. If concentration of A is increased to three times, how will it affect the rate of formation of B?
Assertion (A): Order of reaction is applicable to elementary as well as complex reactions.
Reason (R): For a complex reaction, molecularity has no meaning.
Which of the following statement is true?
