Topics
The Living World: Adaptations and Classification
- Biodiversity
- Adaptations and Its Types
- Adaptations of Plants
- Adaptation in Aquatic Plants (Hydrophytes)
- Adaptation in Desert Plants (Xerophytes)
- Adaptation in plants of snowy regions
- Adaptation in Forest Plants
- Adaptation in Grassland Plants (Mesophytes)
- Adaptation for Ingestion of Food in Plants
- Adaptation in Animals
- Adaptation in Aquatic Animals
- Adaptation in Forest and Grassland Animals
- Adaptation in Desert Animals
- Adaptation in animals of snowy regions
- Adaptation in Aerial Animals
- Adaptation in Reptiles
- Adaptation for Food in Animals
- Adaptation for Blending with the Surroundings
- Classification of Living Organisms
- Taxonomic Hierarchy of Living Organisms: Unit of Classification
- Nomenclature
Plants: Structure and Function
Properties of Natural Resources
Nutrition in Living Organisms
- Nutrients and Nutrition
- Autotrophic Plants
- Symbiotic Plants
- Heterotrophic Plants
- Insectivorous Plants
- Saprophytic Plants
- Role of nutrients and effects of their deficiency on plants
- Transport System in Plants
- Nitrogen Fixation
- Nutrition in Animals
- Mode of Nutrition in Animals
- Holozoic Nutrition
- Saprozoic Nutrition
- Parasitic Nutrition
Food Safety
Measurement of Physical Quantities
Motion, Force and Work
Static Electricity
Heat
Disaster Management
Cell Structure and Micro-organisms
- Cell: Structural and Functional Unit of Life
- Measurement and observation of cells
- Plant Cell and Animal Cell
- Structure of the Cell
- Cell Wall - “Supporter and Protector”
- Plasma Membrane
- Cytoplasm - “Area of Movement”
- Nucleus - “Brain” of the Cell
- Endoplasmic Reticulum (ER)
- Golgi Apparatus - "The delivery system of the cell"
- Lysosome - “Suicidal Bag”
- Mitochondria - “Power House of the Cell”
- Non-living Substances Or Cell Inclusion
- Plastids
- Microorganisms (Microbes) and Microbiology
- Useful micro-organisms
- Harmful Microorganisms
- Pathogens: Disease-producing Micro-organisms
The Muscular System and Digestive System in Human Beings
- Muscular System
- Muscles and Its Types
- Human Digestive System
- The Mouth and Buccal Cavity
- The Teeth and Its Structure
- The Salivary Glands
- The Food Pipe/Oesophagus
- Pharynx/Throat
- The Stomach
- The Small Intestine
- Pancreas
- Liver
- The Large Intestine
- Important Glands of the Digestive System
- Effects of Tobacco, Alcohol, Smoking, on the Digestive System
Changes – Physical and Chemical
- Changes-Physical and Chemical
- Classification of Change: Natural and Man-made Changes
- Classification of Change: Harmful and Useful Changes
- Classification of Change: Slow and Fast Changes
- Classification of Change: Reversible and Irreversible Changes
- Classification of Change: Periodic and Non-periodic Changes
- Classification of Change: Physical Changes
- Classification of Change: Chemical Changes
- Corrosion of Metals
Elements, Compounds and Mixtures
Materials We Use
Natural Resources
Effects of Light
Sound: Production of Sound
Properties of a Magnetic Field
In the World of Stars
- Introduction
- Experiment
Introduction:
Liquids, like solids, expand when heated and contract when cooled. When a liquid is heated, the molecules move faster and spread apart, increasing the liquid's volume. When cooled, the molecules slow down and come closer, causing the liquid to shrink or contract. This behaviour can be observed in everyday life, such as mercury or alcohol thermometers, where the liquid level rises with heat and drops when it cools.
Experiment
1. Aim: To observe how a liquid expands when heated and contracts when cooled.
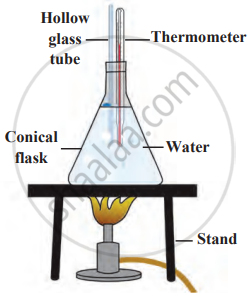
2. Requirements: 500 ml conical flask, two-holed rubber stopper, glass tube, thermometer, measuring ruler, burner, wire gauze, stand, and graph paper.
3. Procedure
- Fill the conical flask with water.
- Insert a glass tube and thermometer into the flask using a rubber stopper.
- Heat the water using a burner and record the water level every 2°C rise in temperature.
- Take 10 readings and observe the water level as it cools down.
- Plot a graph of water level vs. temperature.
Expansion and contraction of a liquid
4. Observation
- As the temperature increases, the water level in the glass tube rises steadily.
- When the water is heated, the particles move faster, and the volume expands.
- After the heating is stopped, the water begins to cool, and the water level gradually drops, showing contraction.

5. Graph: The graph above shows the relationship between temperature and water level. As the temperature rises, the water level increases, demonstrating the expansion of liquids. When the temperature decreases, the liquid contracts. This experiment confirms that liquids expand when heated and contract when cooled.
6. Conclusion: Liquids expand when heated as their particles move apart, increasing volume. Liquids contract when cooled as particles come closer, reducing volume. This experiment demonstrates that temperature changes affect the volume of liquids, with expansion on heating and contraction on cooling.