Topics
Living World and Classification of Microbes
Health and Diseases
Force and Pressure
- Force
- Types of Force: Contact Force
- Types of Force: Non-Contact Force
- Balanced and Unbalanced Forces
- Inertia and Mass
- Types of Inertia
- Thrust and Pressure
- Pressure on Solids
- Pressure of liquid
- Gas Pressure
- Atmospheric Pressure
- Buoyancy Force (Upthrust Force)
- Archimedes Principle
- Density of substance and Relative density
Current Electricity and Magnetism
Inside the Atom
Composition of Matter
- Matter (Substance)
- Characteristics of Particles (Molecules) of Matter
- States of Matter
- The Solid State
- The Liquid State
- The Gaseous State
- Elements
- Types of Element: Metals
- Types of Element: Non-metal
- Type of Element: Metalloid
- Compound
- Types of Compound
- Mixture
- Types of Mixtures
- Solution
- Suspension Solution
- Colloidal Solution
- Molecular Formula of Compounds
- Valency
Metals and Nonmetals
- Types of Element: Metals
- Physical Properties of Metals
- Chemical Properties of Metal
- Types of Element: Non-metal
- Physical Properties of Non-metal
- Types of Element: Non-metal
- Chemical Properties of Non-metal
- Type of Element: Metalloid
- Uses of metals and nonmetals
- Nobel Metal
- Purity of Gold
- Corrosion of Metals
- Alloy
Pollution
- Pollution and Its Types
- Air Pollution and Its Causes
- Effects of Air Pollution
- Prevention of Air Pollution
- Water Pollution and Its Causes
- Effects of Water Pollution
- Prevention of Water Pollution
- Soil Pollution and its Causes
- Effects of Soil Pollution
- Prevention of Soil Pollution
- Relationship of Soil Pollution with Air and Water Pollution
- Laws for Control, Regulation, and Prevention of Pollution by Indian Government
Disaster Management
Cell and Cell Organelles
- Cell: Structural and Functional Unit of Life
- Prokaryotic and Eukaryotic Cell
- Plant Cell and Animal Cell
- Structure of the Cell
- Cell Wall - “Supporter and Protector”
- Plasma Membrane
- Cytoplasm - “Area of Movement”
- Nucleus - “Brain” of the Cell
- Endoplasmic Reticulum (ER)
- Golgi Apparatus - "The delivery system of the cell"
- Lysosome - “Suicidal Bag”
- Mitochondria - “Power House of the Cell”
- Plastids
- Non-living Substances Or Cell Inclusion
Human Body and Organ System
- Human Body
- Human Organ System
- Mechanism of respiration-Breathing
- Human Respiratory System
- Blood Circulatory System in Human
- Human Heart
- Blood Vessels
- Circulation of Blood in the Heart (Functioning of Heart)
- Heart Beat - Heart Sounds "LUBB" and "DUP"
- Blood
- Composition of Blood: Plasma (The Liquid Portion of Blood)
- Composition of Blood: Red Blood Cells (Erythrocytes)
- Composition of Blood: White Blood Cells (Leukocytes)
- Composition of Blood: Blood Platelets (Thrombocytes)
- Functions of Blood
- Blood Transfusion and Blood Groups (ABO and Rh system)
- Blood Pressure (B.P.)
- Heart Related Conditions
Introduction to Acid and Base
Chemical Change and Chemical Bond
Measurement and Effects of Heat
Sound
Reflection of Light
Man Made Materials
Ecosystems
Life Cycle of Stars
- Experiment
- Types of Compound
Experiment
1. Aim: To observe the behaviour of various compounds when heated and distinguish between organic and inorganic compounds based on residue formation.
2. Requirements
- Apparatus: evaporating dish, tripod stand, burner.
- Chemicals: camphor, washing soda, blue vitriol, sugar, glucose, urea.
3. Procedure
- Set up the tripod stand and place the evaporating dish on it.
- Take a small amount of camphor in the evaporating dish and heat it strongly with a burner. Observe what happens to the camphor and note if any residue is left.
- Repeat the process using other compounds: limestone, washing soda, blue vitriol, sugar, glucose, and urea. Carefully observe and record whether any residue is left behind.
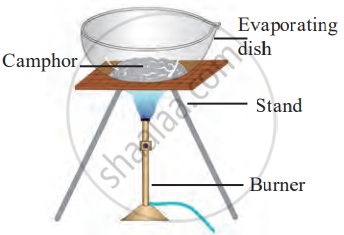
Experimental figure
| Powder in the evaporating dish | Whether there was a residue / no residue in the evaporating dish | Colour of the residue |
|---|---|---|
| Camphor | No residue | None |
| Limestone | Residue present | White |
| Washing soda | Residue present | White |
| Blue vitriol | Residue present | Blue |
| Sugar | Residue present | Black (carbon) |
| Glucose | Residue present | Black (carbon) |
| Urea | Residue present | White |
4. Conclusion: The experiment shows that organic compounds (like camphor, sugar, glucose, and urea) either do not leave a residue or leave a black carbon residue when heated, indicating incomplete combustion. In contrast, inorganic compounds (like limestone, washing soda, and blue vitriol) decompose and leave a residue without forming carbon.
Types of compound:
| Type of Compound | Organic Compounds | Inorganic Compounds | Complex Compounds |
| Definition | Made primarily of carbon atoms bonded to hydrogen, oxygen, and other elements. Known as carbon compounds. | Do not primarily consist of carbon; made from various elements, often forming simple or mineral-based structures. | Also called coordination compounds, they have a complex structure with a metal atom at the centre, surrounded by other atoms or groups. |
| Behaviour on Heating | May not leave a residue or might leave a blackish residue made of carbon due to incomplete combustion. | Decompose, leaving a residue. Do not form blackish carbon residues. | Not specified. Typically more stable due to strong bonds. |
| Examples | Carbohydrates (sugar, glucose), Proteins, Hydrocarbons (petrol, cooking gas), Camphor, and Urea | Common Salt (Sodium Chloride), Soda (Sodium Carbonate), Rust (Iron Oxide), Blue Vitriol (Copper Sulfate), Limestone (Calcium Carbonate) | Chlorophyll (contains magnesium), Hemoglobin (contains iron), and cyanocobalamin (Vitamin B-12, contains cobalt) |
|
Properties/ Structure |
Combine with oxygen to form gases; if combustion is incomplete, carbon residue remains. | High melting and boiling points; do not burn easily. | Metal ions bonded to surrounding atoms in a structured arrangement; strong chemical bonds. |
| Applications /Significance | Essential in biological systems, found in fuels, plastics, and everyday materials. | Used in construction, manufacturing, and industrial processes. | Vital in biological systems and used in industrial applications, catalysts, and medicine. |
If you would like to contribute notes or other learning material, please submit them using the button below.
