Acids and bases dissociate in water, releasing H⁺ ions (from acids) and OH⁻ ions (from bases) in varying proportions. The ratio of H⁺ to OH⁻ ions determines whether a solution is acidic, neutral, or basic.
For example:
- Soil can be classified as acidic, neutral, or basic based on its ion proportions.
- Biological systems like blood or cell sap require specific ion ratios for proper functioning.
- Chemical and biochemical processes, such as fermentation, depend on maintaining specific ion concentrations.
\[\mathrm{H_2O}\xrightarrow{\text{dissociation}}\mathrm{H^++OH}\]
Pure water dissociates slightly to produce equal concentrations of H⁺ ions and OH⁻ ions, with [H⁺] = 1 x 10⁻⁷ mol/L at 25°C. Different solutions have varying H⁺ ion concentrations:
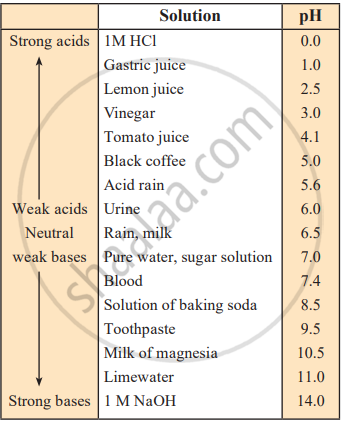
- In 1M HCl, [H⁺] = 1 x 10⁰ mol/L, making it strongly acidic.
- In 1M NaOH, [H⁺] = 1 x 10⁻¹⁴ mol/L, making it strongly basic.
The range of H⁺ ion concentration in aqueous solutions spans from 10⁰ to 10⁻¹⁴ mol/L.
In 1909, Danish scientist Sorensen introduced the pH scale to conveniently express H⁺ ion concentrations. The pH scale ranges from 0 to 14:
- pH 7 represents a neutral solution (e.g., pure water, where [H⁺] = 1 x 10⁻⁷ mol/L).
- pH < 7 indicates an acidic solution.
- pH > 7 indicates a basic solution.
The pH scale is widely used in chemistry and biology to analyse and regulate solutions.