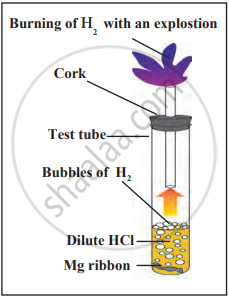
Reaction of Acids with Carbonates and Bicarbonates
1. Aim: To study the reaction of acids with carbonate and bicarbonate salts of metals and confirm the release of carbon dioxide gas.
2. Requirements: Baking soda (sodium bicarbonate, NaHCO₃), lemon juice, limewater (Ca(OH)₂ solution), test tubes, rubber cork with a bent tube, washing soda (sodium carbonate, Na₂CO₃), vinegar, dilute HCl, and dropper.
3. Procedure:
- Take some limewater in a test tube and keep it ready for observation.
- In another test tube, add a small amount of baking soda (NaHCO₃).
- Pour a few drops of lemon juice into the test tube containing baking soda. Immediately, fit the test tube with the rubber cork connected to the bent tube.
- Insert the other end of the bent tube into the limewater and observe.
- Repeat the experiment using washing soda (Na₂CO₃) and replace lemon juice with vinegar or dilute HCl. Record your observations in each case.
4. Observations: Effervescence is observed in the test tube containing baking soda or washing soda when acid is added, indicating the release of a gas. The limewater connected via the bent tube turns milky when exposed to the gas, confirming the presence of carbon dioxide.
5. Conclusion: The reaction between acids and carbonates/bicarbonates of metals produces carbon dioxide gas, which turns limewater milky. The reaction also produces salt and water.
This reaction can be represented by the following chemical equation:
Ca(OH)₂ (aq) + CO₂ (g) → CaCO₃ (s) + H₂O (l)
Carbonate Salt of Metal + Dilute Acid → Another salt of metal + Carbon Dioxide
CaCO₃ (s) + 2 HNO₃ (aq) → Ca(NO₃)₂ (aq) + CO₂ (g) + H₂O (l)
Bicarbonate salt of metal + dilute acid → Another salt of metal + carbon dioxide + water
KHCO₃ (s) + HNO₃ (aq) → KNO₃ (aq) + CO₂ (g) + H₂O (l)